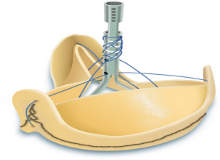

Sorin's Solo Smart aortic pericardial heart valve gets FDA approval

The US Food and Drug Administration (FDA) granted approval for Italy-based medical company Sorin's Solo Smart aortic pericardial heart valve.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The company claims that Solo Smart is the first aortic valve with a removable stent to be approved in the US, providing native-like performance with the ease of implant of a stented valve.
Designed from bovine pericardium, Sorin claims that Solo Smart is fully biological as it does not have any synthetic material. This bioprosthesis is similar to the healthy native aortic valve, which preserves the aortic root physiology.
NICE recommends intrabeam radiotherapy for breast cancer

The National Institute for Health and Care Excellence (NICE) in the UK recommended intrabeam radiotherapy as a treatment option for people with early breast cancer.
In new draft guidance, NICE said that the radiotherapy treatment should be recommended for National Health Service (NHS) funding for its use in early breast cancer, provided patients are properly informed about its pros and cons and that further data are collected.

US Tariffs are shifting - will you react or anticipate?
Don’t let policy changes catch you off guard. Stay proactive with real-time data and expert analysis.
By GlobalDataAccording to NICE, the new treatment has the potential to be a much more efficient form of radiotherapy.
New stem cell operation to revolutionise knee cartilage treatment
Surgeons at the Southampton General Hospital carried out a new knee operation that could prevent the development of arthritis.
During the operation, surgeons used a 'new' knee cartilage from stem cells in hip with special glue, to apply as coating on the damaged cartilage.
Cartilage is a tough, flexible tissue that covers the surface of joints and allows bones to slide over one another that helps in reducing friction as well as acts as a shock absorber.
Boston Scientific's Rebel coronary stent system gets approval from FDA
The US Food and Drug Administration (FDA) approved US-based Boston Scientific's Rebel platinum chromium coronary stent system used for treatment of coronary artery disease (CAD).
The Rebel stent system is the company's latest generation bare-metal stent and expands its family of stents featuring platinum chromium alloy and a customised stent architecture design.
Boston Scientific said that the Rebel stent provides the identical stent platform as the Promus Premier drug-eluting stent but without the Everolimus drug.
St Jude Medical's FlexAbility ablation catheter gets European approval

St Jude Medical received CE Mark approval for its FlexAbility ablation catheter, a new ablation technology used by electrophysiologists, to treat cardiac arrhythmias.
The approval of next-generation ablation catheter further strengthens the company's ablation technology portfolio, which also includes the TactiCath contact-force sensing irrigated ablation catheter.
St Jude Medical has also announced the first use of the FlexAbility ablation catheter that combines an irrigated flexible catheter tip with a new design and handle.
Covidien introduces Emprint ablation system to treat soft tissue tumours
Ireland-based Covidien introduced its Emprint ablation system with Thermosphere technology, designed to precisely heat and destroy diseased soft tissue (including liver, lung and kidney), and non-resectable liver tumours.
The company said that the new ablation system provides physicians predictable results regardless of target location, tissue type or changes in tissue properties during a procedure.
The Emprint ablation system offers clinicians three kinds of spatial energy control including thermal, field and wavelength, to create predictable and spherical ablation zones.
FDA approves launch of Titan's Endoskeleton TL device for lateral spinal fusion

The US Food and Drug Administration (FDA) granted approval to Titan Spine to commercially launch its Endoskeleton TL system, a spinal fusion solution that uses a lateral approach.
Endoskeleton TL is the first device to feature surface technology that is designed to participate in the fusion process by creating an osteogenic response to the implant's topography.
The new device uses the company's roughened titanium surface technology to the lateral approach.
AngioScore introduces new AngioSculpt scoring balloon catheters to treat PAD
Spectranetics's US-based subsidiary AngioScore introduced its new 200mm length AngioSculpt PTA scoring balloon catheters to treat peripheral artery disease (PAD) above-the-knee (ATK).
The US Food and Drug Administration (FDA) granted 510(k) approval to the new AngioSculpt catheters to be marketed for dilatation of lesions in the iliac, femoral, iliofemoral, popliteal, infra-popliteal, and renal arteries, as well as to treat obstructive lesions of native or synthetic arteriovenous dialysis fistulae.
The company said that the new catheters are not approved for use in the coronary or neuro-vasculature.





