Strokes occur when the blood supply to the brain gets cut off and require urgent treatment to remedy. The aftereffects can often be pretty gruelling, with patients often confined to bed for weeks as they recover. This lack of motion leads to reduced blood flow through the legs, which can cause blood clots. Pressure ulcers might also form on the legs, with reduced blood flow making them hard to heal.
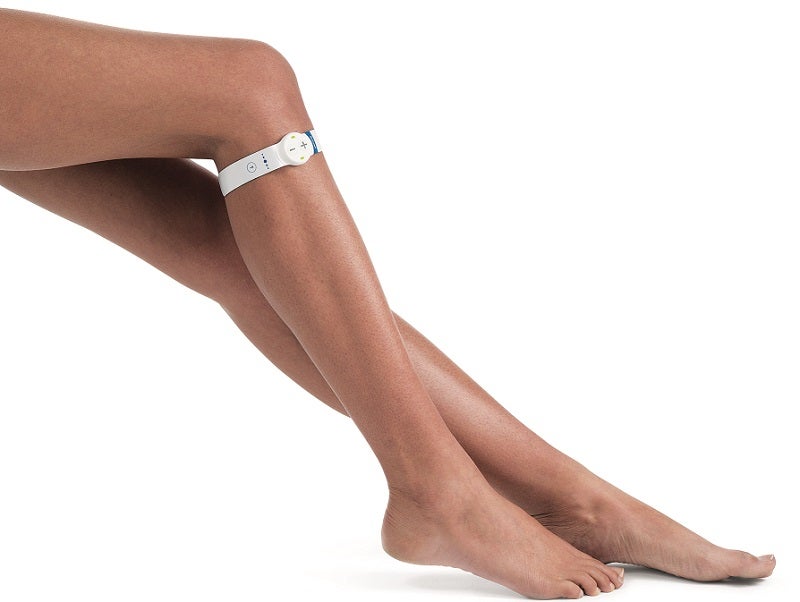
UK-based medtech company Sky Medical Technology has developed an electrostimulation device with patients like these in mind. The company was recently crowned the winner of the Innovation Award at the Medilink North of England Healthcare Business Awards 2020 for its work on the device, known as the geko.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
It had partnered with the NHS Royal Stoke University Hospital to study the impact of the geko on immobile acute stroke patients, and found that patients who wore the device experienced zero incidences of venous thromboembolism (VTE), a common complication of being bedbound. VTE is estimated to cause 40,000 deaths a year in England alone, 62% of which are thought to be preventable.
This study changed clinical practice at Royal Stoke, drove wider NHS adoption of the device across multiple stroke units and led to Sky Medical Technology receiving US Food and Drug Administration (FDA) 501(k) clearance. Today, geko is used by more than ten NHS Trusts, and it’s the only FDA approved device for use on all surgical and non-surgical patients for deep vein thrombosis (DVT) prevention.
How does the geko work?
The geko uses a mechanism of bioelectronic neuromuscular electrostimulation to send a small electrical current through the common peroneal nerve, which is located in the leg at the side of the knee. The stimulation occurs once a second, sending a signal down the nerve to the muscles and prompting them to squeeze against the veins in the leg, pushing blood through and back towards the heart.
Sky Medical Technology CEO Bernard Ross says: “It’s essentially what we do when we walk. The heart pushes oxygenated blood around the body, and when we walk the muscles squeeze against the veins in the calf, foot and to some degree in the thigh – that pushes blood back towards the heart. The geko is able to trigger 62% of the venous return, aka the blood flow back to the heart, that you get when you walk, which is helpful for a number of indications.”
This is able to prevent the formation of blood clots in the veins of the leg. By keeping the blood flow going through the veins, clots are unable to form.
Ross says: “That increased blood flow, because it increases venous return, empties blood from the capillary bed. This means each squeeze of the heart has less resistance in the capillaries, meaning you increase arterial blood flow as well. This then means that you get much more oxygenated blood in the skin, particularly in the feet and in the legs.”
The increased blood flow means that if a patient has wounds that aren’t healing due to poor circulation – venous leg ulcers, arterial ulcers, diabetic ulcers – then geko can help the ulcers heal.
Keeping the blood circulating when people can’t do it themselves also allows the absorption of intertissue fluids into the circulatory system, which can reduce postsurgical and trauma swelling
Ross says: “If someone has a broken ankle, the ankle swells. If you want to do an operation on them you have to wait until the swelling has gone down, which can take five to seven days. When you apply the geko device when someone’s got a broken ankle, you can generally operate the next day.”
The device needs to be disposed of and replaced each day, which may make it seem like it’s not the most environmentally friendly option in the world. But when the current alterative is external compression, and all the batteries, plastic and disposable leggings that come with it, geko comes out surprisingly green.
Ross says: “This technology, even though it’s replaced daily, has a twentieth of the environmental impact of comparable products. You have within it plastics, hydrogel, a printed circuit board and the battery, and all of these items can be recycled. In fact, we believe something like 70% of the plastics involved in this product have already been recycled.”
geko has also been trialled in other indications
Clinical trials have taken place into other indications too. Canadian researchers have looked at the use of the geko device to reduce oedema after kidney transplant, and found that geko was able to significantly reduce post-surgical swelling in the legs. Urinary output also doubled in the first five days in the geko-wearing patients, indicating that the function of the implanted kidney improved much faster when the geko was worn during recovery.
Sky Medical Technology also has data which suggests that in congestive heart failure patients, coronary artery flow is increased by 15%. The company has also seen increased blood flow as high up as the back of the hand in patients with venous insufficiency at all levels.
Further study will need to be carried out for all of these indications before the device can be used for them in the clinic. The company is now investing funds in developing its clinical data, in the hope of becoming increasingly attractive to larger companies.
Ross says: “What we’d like to do is actually have the opportunity to build up the value of the business more before this leads to an exit, but ultimately that will be a shareholder decision.
“We do see the potential of this to become a very important technology. Clinicians are saying to us that they can see the use of the geko device becoming ubiquitous in healthcare. It could become part of recovery for almost every hospital based procedure.”



