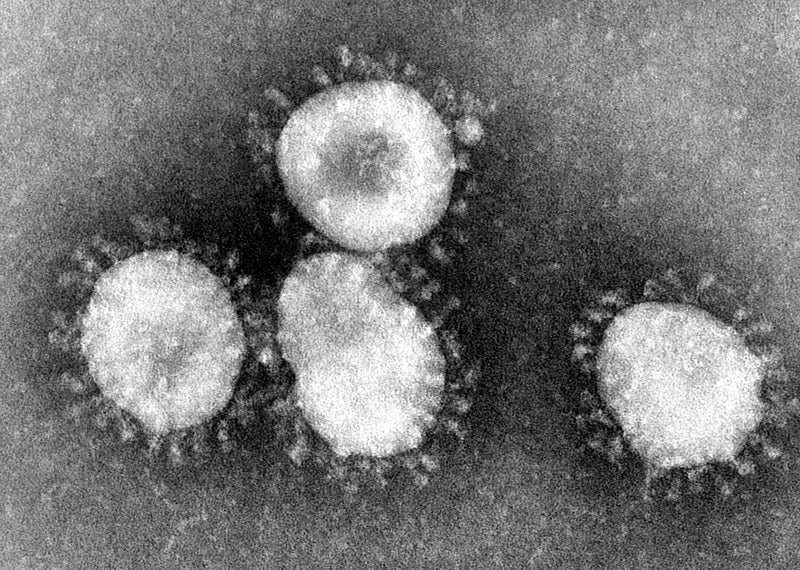
Primerdesign launches molecular test for new coronavirus
Clinical diagnostics company Novacyt’s molecular diagnostics division Primerdesign launched a molecular research use only (RUO) test for new coronavirus (2019-nCoV).
The company developed the test in response to the recent coronavirus outbreak in China.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Primerdesign’s coronavirus test is capable of detecting only the 2019 strain of the virus.
Study reveals radiotherapy can cure bladder cancer
A report published by The Institute of Cancer Research (ICR) revealed that bladder cancer extended to the lymph nodes of the pelvis can be treated through targeted forms of radiotherapy.
Scientists at ICR London carried out a phase II clinical trial evaluating the use of radiotherapy in patients with bladder cancer, which had extended to the pelvic lymph nodes.
The study was conducted in collaboration with researchers and clinicians at The Royal Marsden NHS Foundation Trust.
US researchers to trial retinal scanning to diagnose Alzheimer’s
Researchers at the University of Rhode Island, Butler Hospital and BayCare Health System collaborated to launch retinal scanning tests for early detection of Alzheimer’s disease.
Morton Plant Mease Health Care Foundation and St Anthony’s Hospital Foundation provided funds for this five year, $5m Atlas of Retinal Imaging in Alzheimer’s Study (ARIAS).
Researchers claimed that the simple retinal screening test could be more cost-effective compared to the current positron emission tomography scans, which can detect amyloid plaque in the brain, associated with Alzheimer’s before symptoms appear.
California-based Mojo Vision developing smart contact lens
California-based Mojo Vision is developing a smart contact lens with a built-in display, providing beneficial patient information to prevent loss of focus.
Under the research and development (R&D) phase, Mojo lens offers Invisible Computing, a platform that provides information instantly, allowing individuals to interact with each other.
For over a decade, Mojo has been holding patents required for the development of an augmented reality (AR) smart contact lens.
FDA clears new AI system to spot stroke
The US Food and Drug Administration (FDA) granted 501(k) clearance to radiology company Aidoc’s AI-based system for the detection of large-vessel occlusion (LVO), ischaemic strokes that result from a blockage in one of the major arteries of the brain.
The clearance marks the company’s fourth FDA-approved AI package and its second for stroke, having previously received clearance for an AI module that flags and prioritises intracranial haemorrhage.
The two stroke modules provide a comprehensive AI package for the identification and triage of both ischaemic and haemorrhagic stokes in computerised tomography (CT) scans.
Healthy.io launches digital wound management solution
Healthy.io, a creator of smartphone urinalysis, launched a digital wound management solution, as an extension to its clinical-grade colour recognition products.
The solution is the company’s second product line, created to assist healthcare professionals in evaluating chronic wounds and track their progress over time.
The company stated that the existing method for measuring and documenting chronic wounds is inconsistent and rudimentary, as nurses make measurements using more basic tools such as paper rules.
Pentax Medical gets FDA approval for IMAGINA endoscopy system
Pentax Medical, a unit of Hoya Group, received 510(k) clearance from the US Food and Drug Administration (FDA) for its IMAGINA endoscopy system.
The endoscopy platform was developed for gastrointestinal (GI) procedures at ambulatory surgery centres (ASC).
IMAGINA’s user interface and endoscope design features will provide practitioners with enhanced visualisation and improved operator experience.
Roche signs 15-year deal with Illumina for cancer diagnostic
Swiss pharmaceutical company Roche entered a partnership with US-based Illumina to provide broad access to clinical oncology next-generation sequencing (NGS).
The 15-year, non-exclusive agreement gives Roche rights to develop and distribute in vitro diagnostic tests on Illumina’s NextSeq 550Dx System, in addition to its upcoming portfolio of Dx sequencing systems.
The partnership will also see Roche joining forces with Illumina to expand the claims for Illumina’s pan-cancer assay TruSight Oncology 500 (TSO 500).
Sebia and Sanofi partner to develop multiple myeloma treatment
In vitro diagnostic company Sebia teamed up with French pharmaceutical firm Sanofi to develop a multiple myeloma diagnostic test.
The test will alleviate possible interference from Sanofi’s investigational antibody, isatuximab in Immunofixation Electrophoresis (IFE) tests.
Isatuximab is a monoclonal antibody targeting a specific epitope of CD38 capable of triggering action that promotes programmed tumour cell death and immunomodulatory activity.
Abbott receives FDA approval for heart pump implant procedure
Abbott secured approval from the US Food and Drug Administration (FDA) for its less-invasive heart pump implant procedure to help patients avoid open-heart surgery.
With the approval, Abbott’s HeartMate 3 heart pump can be implanted in patients with advanced heart failure through an incision in the chest wall.
The device uses Full MagLev technology to reduce trauma to blood passing through the pump and improve outcomes for patients.





