During this year’s Migraine Awareness Week, which runs from 2-8 September, the struggle to find successful pain relief treatments is back under the spotlight. US-based company electroCore thinks it has the answer that many migraine sufferers are desperately looking for. Its gammaCore device is a new non-invasive treatment that targets the vagus nerve but is currently only available when authorised by specialists.
In the UK, electroCore is working with the NHS and NICE on reimbursement agreements so that gammaCore can be made available to more cluster headache patients. It is provided to patients for 93 days free of charge before its effects are assessed, after which treatment costs £750 every three months. So far the device has been effective in 50% of patients so is it worth the investment? electroCore’s chief medical officer Peter Staats explains all about gammaCore and of its impact on migraine and cluster headache pain relief.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Charlotte Edwards: Tell me about electroCore and your gammaCore device?
Peter Staats: Electrocore has been around for almost 15 years. We founded the company back in 2005 with the goal of stimulating the vagus nerve to see what therapy effects we could see. We’ve studied a variety of different areas over the last 15 years and electroCore’s primary product now is a non-invasive approach to stimulating the vagus nerve.
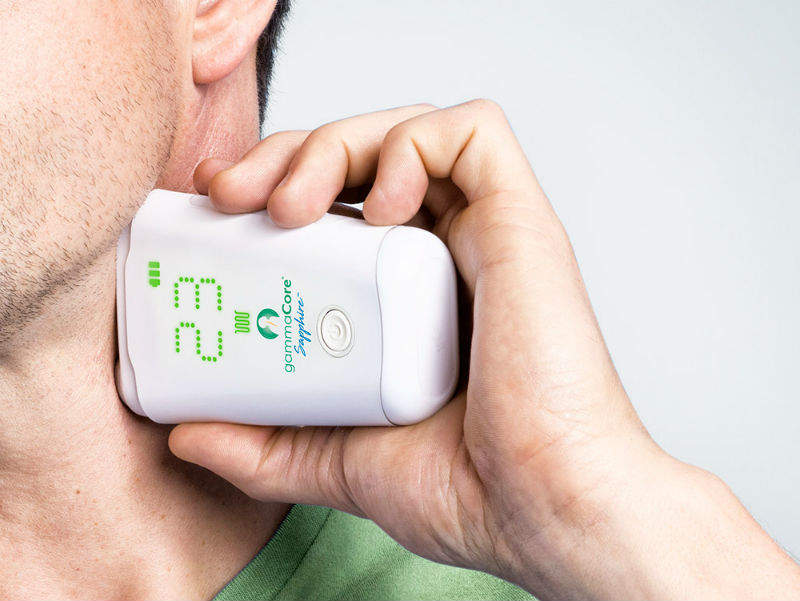
We are the first to take this approach of stimulating the vagus nerve at the neck non-invasively. Prior to us, if one wanted to stimulate the vagus nerve then they would have to implant an electrode and connect it to a pulse generator underneath the skin. One of our innovations has been developing a technique that takes this out of the hands of the surgeon and really into the hands of the patient.
The patient controls their own ability to stimulate the vagus nerve with a hand-held device the size of a small cell phone. They put a bit of conducting gel on the neck, they turn on the device and it gives you stimulation for about two minutes and there are a variety of health benefits that have been noted with that. In the United States we’re approved for episodic cluster headache and for the acute treatment of migraines. In Europe we also have broader indications in psychiatry, respiratory disease, and gastro diseases as well.
CE: Why target the vagus nerve in the treatment of migraine and cluster headaches?
PS: In my former life I was an anaesthesiologist and the vagus nerve was simply that structure that I knew to avoid but didn’t really know what it did. It turns out the vagus nerve is incredibly important. When you think about how we developed as a species, original information transfer between the brain and the gut was through the prehistoric vagus nerve. Over time it has persisted that the vagus nerve really is, what I will term, the mind-body interface.
When I started with this company many, many years ago we started looking at shock and turning off shock in a variety of different animals to demonstrate that when an animal is bleeding out their life can be saved by stimulating the vagus nerve. We then progressed to study airway reactivity and asthma and then as we were doing this a variety of developments came forward.
We were doing some studies with asthma patients in Africa and asked, as most scientists do, “Are you feeling anything else?” The patients started to note that their headaches went away. So we actually stopped to pause and looked at the literature and in the literature there are reports of people with vagus nerve implants for epilepsy who had reported that their headaches had gotten better. So, we did a variety of studies and worked collaboratively to study headaches. We spent many years understanding what happens with migraines and cluster headaches and figured out that we could stimulate the vagus nerve non-invasively to control mechanistically what happens with a headache. Then we did a variety of studies which found that this is an effective therapy for a variety of different headaches.
CE: When the device is stimulating the vagus nerve, what is happening to make migraine pain go away?
PS: There are a variety of things that occur. First, the vagus nerve works on modifying neurotransmitters in the brain and one of the main neurotransmitters is one called glutamate. Glutamate is the most prevalent excitatory neurotransmitter in the brain and it is produced significantly during a migraine attack. We believe neurotransmitters have a major impact and some of the physicians at Harvard used our device to show its effect on critical depression and the associated waves of electrical activity which worked similar to drugs at normalising these waves but much quicker.
Secondly, [during a migraine] there are areas of the brain which start to send electrical signals, what we call sensitivity of the brain, and these can be upregulated but vagus nerve stimulation can bring that right back down to being normal again. In addition, we believe there are changes of the supporting structures of the brain that give a longer term effect. We’re studying that right now at Harvard.
For a small company like ours, we’re investing a tremendous amount in understanding the science of how our therapy works. We’re spending money and resources demonstrating that it does work at level 1 studies and I believe we have the only level 1 study of migraines with a non-invasive neuro device.
CE: So which patients can go to their doctor now and ask to be prescribed this device?
PS: So right now in the UK I believe any physician can prescribe it but it’s primarily going to be headache disorder and neurology specialists and they can prescribe it for the patient.
CE: How safe is gammaCore for migraine patients to use at home?
PS: We don’t actually know of any adverse safety problems with using this device a lot when compared to other therapies out there. For example, cluster headache patients can give themselves injectable triptans and they have cardiac warnings; we don’t have any of those warnings. In the US people can only get up to eight injectable triptans a month whereas patients can use our device 24 times a day. So the safety profile is really unparalleled with what we’re doing.
We are optimising the body’s own ability to control problems so we’re not doing something that we think is unsafe. The regulators and others have wanted to be very sure of that of course. The vagus nerve does have innervation on the heart so we’ve done all kinds of studies to demonstrate that we don’t stop the heart, we don’t cause ischaemia, we don’t cause bronchial decline. There are a small number of people who have skin irritation, that would be the biggest problem that we see, but this is actually pretty rare.
CE: Will the device be expensive?
PS: [We’ve taken] what is otherwise a very expensive device and decided that we’re only going to bill patients if they are doing well when they’re using it. Our device, like any other device, might not work well for everybody. So we have a monthly bill like a pharmaceutical agent.
We’re making sure we really do help patients because if we do all this and we don’t have patients cared for then we’ve done nothing. We believe we’re really cost-effective too, unlike some of the new strategies that are coming out across healthcare. We believe we are cost-effective if not cost-saving for the system.
CE: What is next for electroCore?
PS: For our next big project we are diving into rheumatology and have a series of studies either ongoing or planned to demonstrate how vagus nerve stimulation can be an important treatment in diseases beyond just headaches, including rheumatological diseases.
I think what we’re seeing is the tip of the sphere of bioelectric medicine. We are figuring out how to access the nervous system non-invasively and we are starting to decode how the nervous system controls disease, tap into it and modify the disease with electricity. I think you’ll see more companies like us follow but we are at the beginning of bioelectric-medicine, we are the pioneers. We’re not alone in this but what we are is the first to bring the therapy to the hands of the patient in a cost-effective method that the patient can control.





