Researchers at the University of California San Diego School of Medicine and Institute of Engineering in Medicine have successfully 3D-printed spinal cord implants which can be loaded with neural stem cells and have the potential to repair spinal cord injuries.
The 3D-printed spinal cord implants have been described in a study published in the latest issue of Nature Medicine. They have been designed to promote nerve growth across spinal cord injuries by restoring connections and lost function. When tested in rat models, the scaffolds were found to support tissue regrowth, stem cell survival and expansion of the neural stem cell axons from the scaffold to the host spinal cord.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Translational Neuroscience Institute at UC San Diego School of Medicine director and co-senior author of the study Professor Mark Tuszynski said: “In recent years and papers, we’ve progressively moved closer to the goal of abundant, long-distance regeneration of injured axons in spinal cord injury, which is fundamental to any true restoration of physical function.”
Co-first author Dr Kobi Koffler added: “The new work puts us even closer to real thing because the 3D scaffolding recapitulates the slender, bundled arrays of axons in the spinal cord. It helps organize regenerating axons to replicate the anatomy of the pre-injured spinal cord.”
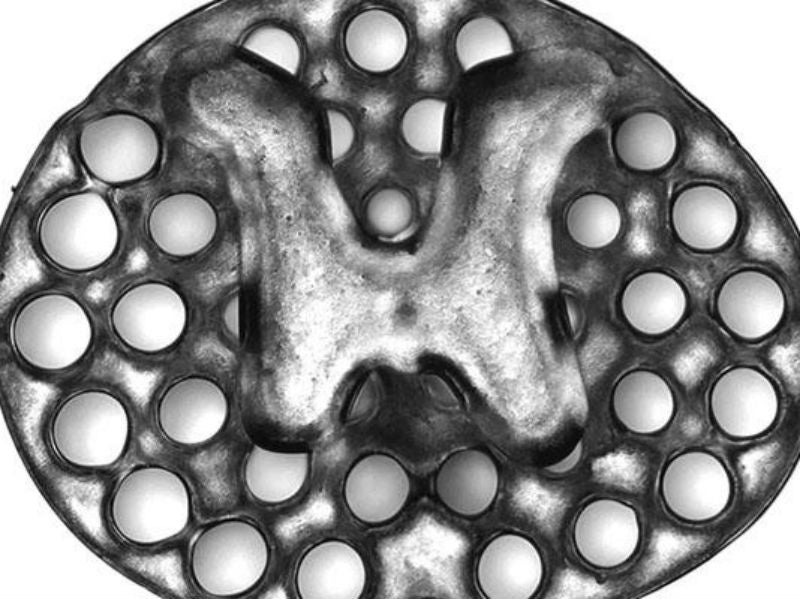
The research team used rapid 3D printing technology to create the scaffold, which mimics central nervous system structures and acts as a guide for new axons to grow in the right direction and complete the spinal cord connection.
The implants consist of dozens of 200-micrometer-wide channels that guide neural stem cell and axon growth along the extent of the spinal cord injury. The printing technology was used by co-senior author of the study Professor Shaochen Chen and his team, who were able to produce two-millimetre-sized implants in 1.6 seconds. In contrast, traditional nozzle printers normally take several hours to produce much simpler structures.
The process has yet to be tested in humans but is scalable to human spinal cord sizes. To prove this, the researchers printed four-centimetre-sized implants modelled from MRI scans of actual human spinal cord injuries. They were able to do this within ten minutes.
In the rat models, the two-millimetre implants with the loaded stem cells were grafted into sites of severe spinal cord injury and aided complete spinal cord tissue regrowth over a few months. The implants were also able to aid the connection of the severed ends of the host spinal cord. The treated rats reportedly regained significant functional motor improvement in their hind legs.
Koffler said: “This marks another key step toward conducting clinical trials to repair spinal cord injuries in people. The scaffolding provides a stable, physical structure that supports consistent engraftment and survival of neural stem cells.
“It seems to shield grafted stem cells from the often toxic, inflammatory environment of a spinal cord injury and helps guide axons through the lesion site completely.”
The circulatory systems of the treated rats were also found to have penetrated inside the implants and formed functioning networks of blood vessels. This helped the neural stem cells survive.
Zhu added: “Vascularisation is one of the main obstacles in engineering tissue implants that can last in the body for a long time. 3D-printed tissues need vasculature to get enough nutrition and discharge waste.
“Our group has done work on 3D-printed blood vessel networks before, but we didn’t include it in this work. Biology just naturally takes care of it for us due to the excellent biocompatibility of our 3D scaffolds.”
The scientists are now working on scaling up the technology so they can test it in larger animal models as preparation for human trials. The next step involves the introduction of proteins within the spinal cord scaffolds, which will increase stem cell survival and axon outgrowth.





