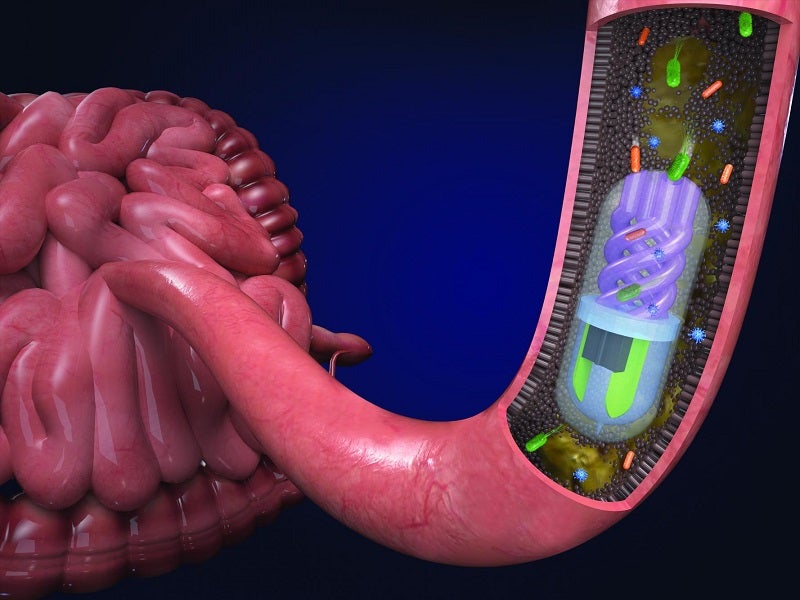
Researchers at Tufts University in the US have developed a 3D printed pill with an osmotic pump which can sample bacteria found in the gut as it passes through the gastrointestinal (GI) tract.
This technology could be used to profile bacterial species inhabiting the gut, helping to diagnose and treat conditions affected by the microbiome.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
This pill represents the first non-invasive diagnostic tool capable of profiling microbiome populations throughout the entire GI tract. Current methods involve analysis of faecal DNA and metabolites, but this provides limited information about the GI environment prior to the distal colon, where bacterial species can vary significantly.
The pill is 3D printed with microfluidic channels which sample different stages of the GI tract. The surface is covered with a pH-sensitive coating so it doesn’t absorb any samples until it enters the small intestine, where the coating dissolves.
Underneath the coating, a semi-permeable membrane separates two chambers inside the pill, with one containing helical channels which take up bacteria and the other containing a calcium salt. The salt chamber helps create an osmotic flow across the membrane, pulling bacteria into the helical channels. A fluorescent dye in the salt chamber helps locate the pill after it exits the GI tract.
The pill can also be used to target certain locations within the gut through a small magnet placed inside. By placing a magnet outside the body, clinicians can hold the pill in place in these areas, should a patient need a specific part of their GI tract tested.

US Tariffs are shifting - will you react or anticipate?
Don’t let policy changes catch you off guard. Stay proactive with real-time data and expert analysis.
By GlobalDataTufts University post-doctoral fellow and lead study author Hojatollah Rezaei Nejad said: “We have incredible technology to analyse bacterial populations using DNA sequencing techniques, but until now have not had a way to sample bacteria throughout the GI tract in a way that was not invasive. By sampling non-invasively, this pill could help us better identify and understand the role of different bacterial species in health and disease.”
Thus far, the pill has only been tested in pigs and primates, and clinical trials will be needed to determine if it can be safely used in humans.





