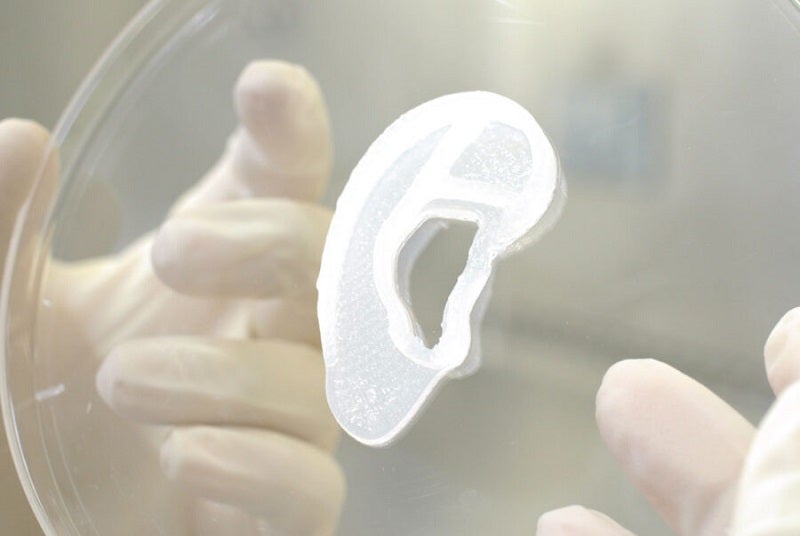
3DBio Therapeutics and the Microtia-Congenital Ear Deformity Institute have conducted a human ear reconstruction using the AuriNovo implant, a 3D-bioprinted living tissue ear implant.
The investigational, 3D-bioprinted implant has been developed using a proprietary process that uses the patient’s own tissue to construct an implantable ear.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Developed using 3D-bioprinting technology, the patient-specific living tissue implant is intended for surgical reconstruction of the outer ear in people who are born with microtia Grades II-IV.
AuriNovo has been designed as an alternative treatment for microtia patients, who have been using rib cartilage grafts and synthetic materials for the reconstruction of the outer ear.
It has received Orphan Drug and Rare Pediatric Disease Designations from the US Food and Drug Administration (FDA).
The reconstructive procedure in the first-in-human Phase I/IIa clinical trial was performed by a team led by Microtia-Congenital Ear Deformity Institute in San Antonio, Texas founder and director Arturo Bonilla.
The trial has been designed to assess AuriNovo’s safety and preliminary efficacy for microtia patients.
AuriNovo will incorporate the own auricular cartilage cells of the patient into a 3D-bioprinted, living, full-sized ear construct after a 3D scan of the geometry of the patient’s opposite ear.
3DBio Therapeutics CEO and co-founder Daniel Cohen said: “This is a truly historic moment for patients with microtia, and more broadly, for the regenerative medicine field as we are beginning to demonstrate the real-world application of next-generation tissue engineering technology.
“It is the culmination of more than seven years of our company’s focused efforts to develop a uniquely differentiated technology platform meeting the FDA’s requirements for therapeutic manufacturing of reconstructive implants.
“We believe that the microtia clinical trial can provide us not only with robust evidence about the value of this innovative product and the positive impact it can have for microtia patients, but also demonstrate the potential for the technology to provide living tissue implants in other therapeutic areas in the future.”
About 11 subjects are expected to be enrolled in the AuriNovo Phase I/IIa clinical trial, which is being conducted in Los Angeles, California and San Antonio, Texas.
Additionally, the company stated that the trial is enrolling patients at Cedars-Sinai Medical Center in Los Angeles, California.





