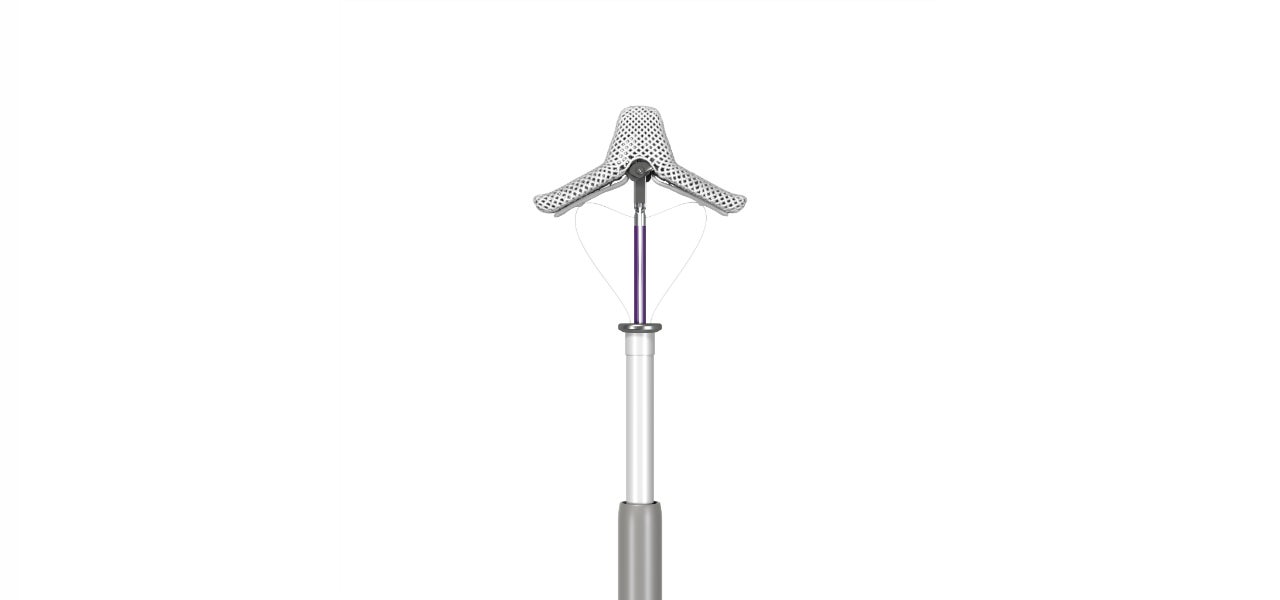
Abbott has received CE Mark for fourth-generation MitraClip Transcatheter Mitral Valve Repair System, its latest minimally invasive mitral valve repair device.
Called MitraClip G4, the device is now approved as a non-surgical option to treat mitral regurgitation (MR) in Europe and other countries that recognise CE Mark. The device is already cleared for use in the US.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The MitraClip G4 system features enhanced clip-based technology, which builds on the MitraClip’s proven delivery system.
It is designed to provide physicians with advanced steering during implantation. Furthermore, the new delivery system has four clip sizes, including two wider clips, offering a variety of treatment options to suit patients’ individual mitral valve anatomy.
The new device also provides independently controlled grippers that will enable physicians to grasp one or both mitral valve leaflets at a time during the MitraClip procedure, if necessary.
Abbott structural heart business senior vice-president Michael Dale said: “An enduring measure of our mission to help people live better lives through better health is our success in advancing new standards of care for the treatment of structural heart diseases.
“This CE Mark, along with other recent approvals and advancements for our MitraClip device, underscores the need for MitraClip’s innovative therapy – which has become a preferred choice for the treatment of mitral regurgitation around the world.”
MR is a common heart condition that affects one in ten adults above the age of 75.
Patients suffering from this progressive condition have a mitral valve that does not close fully, allowing blood to flow backward into the left atrium of the heart instead of flowing forward and to the rest of the body.
Typically, open-heart surgery is considered as the preferred treatment method for MR. However, it is not applicable to all patients due to the potential risk of complications, stemming from comorbidities, advanced age or other issues.
MitraClip is considered to be the first-of-its-kind minimally invasive transcatheter mitral valve repair (TMVr) therapy that can be a life-saving treatment option for select patients with primary or secondary MR.
It is delivered to the heart through a vein in the leg. It reduces the backflow of blood by clipping together portions of the leaflets, or flaps, of the mitral valve.
Once in place, the device restores the normal functioning of the mitral valve and the heart’s ability to pump oxygenated blood.
The company noted that MitraClip has over 16 years of clinical experience with proven safety, survival and durable clinical outcomes.
The clinical study of more than 1,000 MitraClip patients found 99% implant success rate. It also demonstrated MR reduction to the level of none or trace in patients with either primary MR or secondary MR at 30 days.





