Abbott has started LIFE-BTK clinical trial to assess the safety and effectiveness of its new Esprit BTK Everolimus Eluting Resorbable Scaffold System.
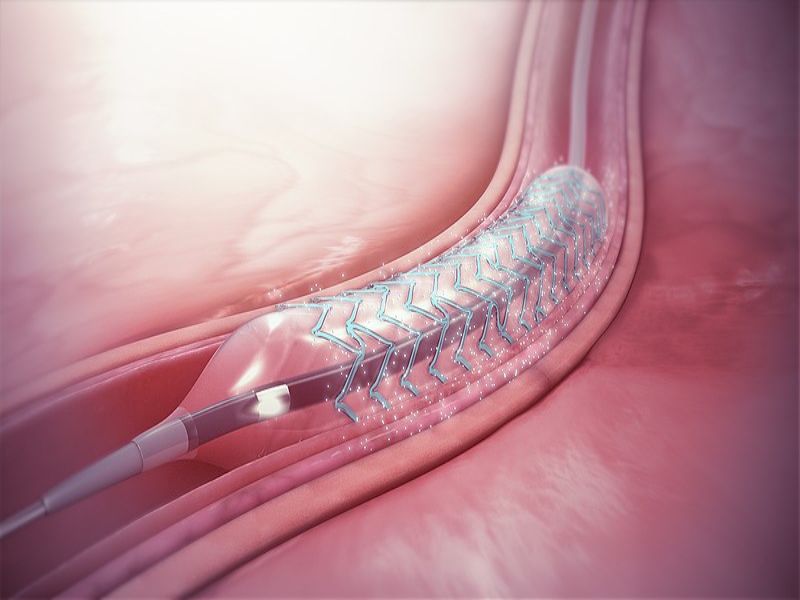
The company’s Esprit BTK System is a semi-crystalline bioresorbable polymer developed to prevent vessel recoil and offer a platform for drug delivery.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
It consists of a thin strutted scaffold, which measures 99 microns made from poly-L-lactide (PLLA), uniformly coated with poly-D, L-lactide (PDLLA) and the cytostatic drug, everolimus.
The LIFE-BTK trial is the first Investigational Device Exemption trial in the US to assess a fully dissolvable device for the treatment of critical limb ischemia in patients with advanced stages of peripheral artery disease (PAD).
Abbott vascular business chief medical officer and Medical Affairs divisional vice-president Nick West said: “Far too many people are impacted by peripheral artery disease, and this new drug-eluting resorbable scaffold is needed to offer meaningful improvements in how this disease is treated.
“Patients treated with balloon angioplasty often require repeat procedures on treated arteries, and therefore, a drug-eluting resorbable device is ideally suited to provide mechanical support for the vessel, reduce the chance of vessel re-narrowing and then gradually disappear over time.”
Unlike traditional metal stents, Abbott’s Esprit BTK System is not a permanent implant.
It is designed to offer support to an artery, following balloon angioplasty, and prevents the vessel from reclosing. The scaffold, once implanted, delivers a drug over a few months to keep the artery open and help its healing.
Over time, it is naturally resorbed into the body, leaving only a healed artery behind.
The Esprit BTK was granted breakthrough device designation by the US Food and Drug Administration (FDA) to streamline its review and pre-market approval timelines.
The first patient to LIFE-BTK trial was enrolled at New York-Presbyterian / Columbia University Irving Medical Center.
Abbott plans to enrol 225 patients at multiple centres around the world. To date, the trial has sites in Australia, Japan, New Zealand, Singapore and the US.
The LIFE-BTK trial builds on the previous studies of the company, examining resorbable technology in treating diseased vessels and blocked arteries.
Long-term clinical data from a meta-analysis of randomised Absorb trials suggested that bioresorbable scaffolds might be an acceptable alternative to metallic DES for many patients with coronary artery disease, the company noted.
Last month, Abbott began participant enrolment into the TactiFlex PAF IDE clinical study of its new catheter device for the treatment of paroxysmal atrial fibrillation (PAF).





