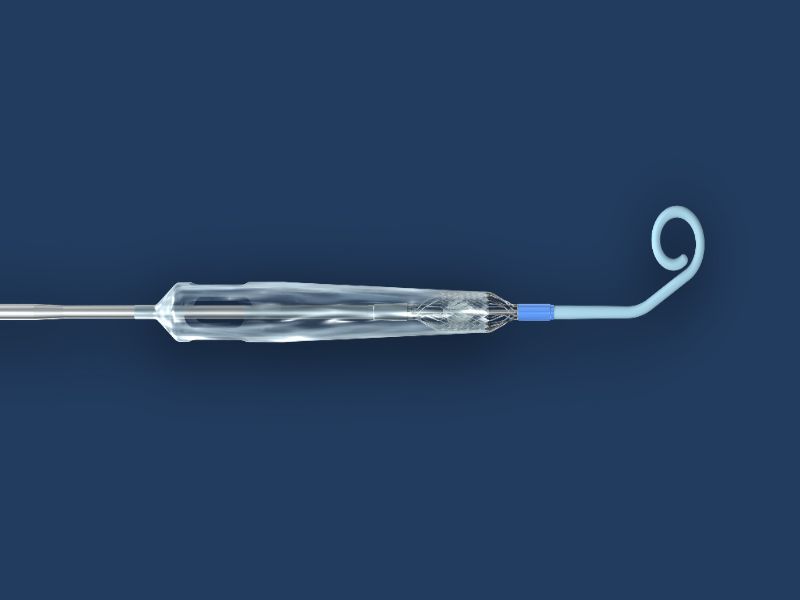
Abiomed has received the US Food and Drug Administration (FDA) investigational device exemption (IDE) approval to begin a clinical trial for the Impella ECP heart pump in high-risk percutaneous coronary intervention (PCI) patients.
Impella ECP is considered to be the world’s smallest heart pump. Delivered through a slender-profile sheath, the device attains peak flows greater than 3.5 L/min. It is un-sheathed in the descending aorta and expands to approximately 18 Fr.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
It also crosses the aortic valve, using a pigtail and pumps from inside the ventricle. The pump is re-sheathed back down to 9 Fr when the procedure is complete.
Furthermore, the pump is designed to sit across the valve with a soft atraumatic polyurethane cannula that opens only when the pump is flowing.
It offers both circulatory support and left ventricular unloading and is intended for short-term mechanical support in patients undergoing a high-risk PCI.
Abiomed chairman, president and CEO Michael Minogue said: “We are excited about the benefits of this new breakthrough technology, which advances the field of heart recovery.
“Abiomed is making significant investments to develop smaller, smarter and more connected technology that forms the foundation of the company’s continued leadership in the field of mechanical circulatory support.”
The prospective, multi-centre, non-randomised trial will assess the safety and feasibility of the device in high-risk PCI patients.
Successful delivery, initiation and maintenance of adequate hemodynamic support are the primary endpoints of the study.
The study will enrol and treat up to five US patients who require revascularisation. Enrolment will begin later this year.
Abiomed also intends to expand the trial to include additional patients later with approval from the FDA.





