A clinical trial being conducted by Stanford University School of Medicine in the US to evaluate the use of Apple Watch for identifying heart-rhythm disorder has signed up more than 400,000 participants.
The Apple Heart Study aims to determine if a mobile app that leverages Apple Watch’s optical sensor to analyse pulse rate data will be able to detect irregular heart rhythms that indicate atrial fibrillation.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Apple and Stanford Medicine carried out the trial enrolment via an iPhone app. The trial will now involve data collection and is expected to be completed early next year.
Stanford University School of Medicine dean Lloyd Minor said: “We hope this study will help us better understand how wearable technologies can inform precision health. These new tools, which have the potential to predict, prevent and manage disease, are finally within our reach.”
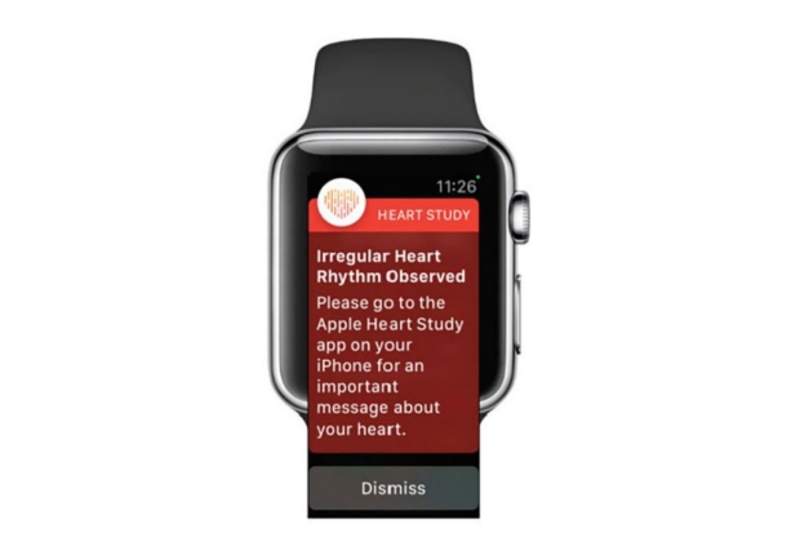
A mobile medical app by Apple captures data from new hardware on the Apple Watch Series 4 to take an electrocardiogram.
The company has also developed a second app that utilises data from an optical sensor embedded in the Apple Watch Series 1 to analyse pulse data for any irregular heart rhythms and alerts the users.
Both the apps secured the US Food and Drug Administration (FDA) clearance in September this year. The Apple Heart Study is evaluating the second medical app.
The participants receiving irregular pulse notifications during the trial will be cross-checked for the presence of atrial fibrillation using ECG patch monitoring.
In addition, the trial will follow this group of subjects to note the proportion that go on to receive medical attention, and also assess the watch’s accuracy to identify irregular pulse.
Apple entered a similar collaboration with healthcare company Zimmer Biomet in October this year to enable the use of Apple Watch and iPhone to help knee and hip replacement surgery patients.





