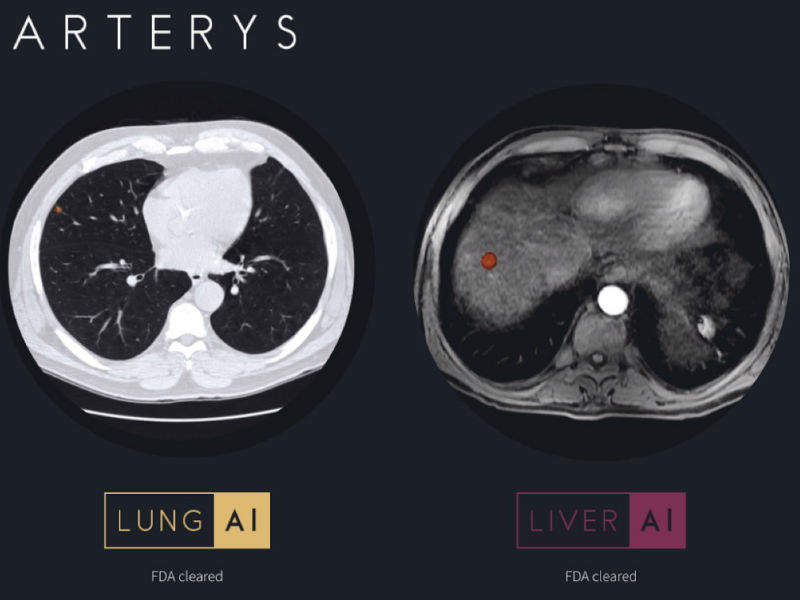
Medical imaging software company Arterys has announced that its Oncology AI suite has received 510(k) clearance from the US Food and Drug Administration (FDA).
The new oncology software uses artificial intelligence (AI) to help clinicians measure and track tumours or potential cancers and easily applying radiological standards.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
It uses deep learning to automate the segmentation of lung nodules and liver lesions, with accuracy equal to segmentations performed manually by experienced clinicians. The clinician has the capability to edit these automated segmentations, so they always remain in control.
The initial deep learning workflows will be for liver MRI and CT scans as well as for lung CT scans. Radiologists can use the technology to easily confirm, evaluate, quantify, and report on the absence or presence of lung nodules and liver lesions along with their key characteristics using a simple web browser. The company plans additional deep learning workflows for solid tumours in other organs.
Arterys co-founder and radiologist, Albert Hsiao said: “The evaluation of primary and metastatic disease in the lung and liver are among the most valuable contributions of radiologists to the care of patients with cancer. We desperately need more efficient technology to automatically track lung and liver lesions to further improve diagnosis, assess response to treatment, and automate reporting with standardised terminology including Lung-RADS and LI-RADS. The Oncology Lung AI and Liver AI products are designed to maximise efficiency and accuracy of the radiologist read and will power next-generation radiology interpretation.”
Oncology AI runs on the Arterys Medical Imaging Cloud AI (MICA platform, which the company claim is easier to deploy than on-premise imaging systems and complies with patient data privacy and security requirements in 27 countries. These countries include the US, Canada, all of Europe, Australia, and New Zealand.
Arterys CEO, Fabien Beckers, PhD, said: “Cancer is one of the leading causes of morbidity and mortality worldwide, with about 14 million new cases in 2012, and about 8.8 million deaths in 2015, according to the World Health Organisation.
“A core Arterys mission is to help the medical community identify cancer earlier, so that patients can receive optimal treatment and improved prognosis. FDA clearance of our Oncology AI suite will help clinicians to quickly measure and track tumours over time. Additionally, our software enables seamless collaboration for clinicians, so second opinions can be easily gathered from within the hospital, or from outside experts.”





