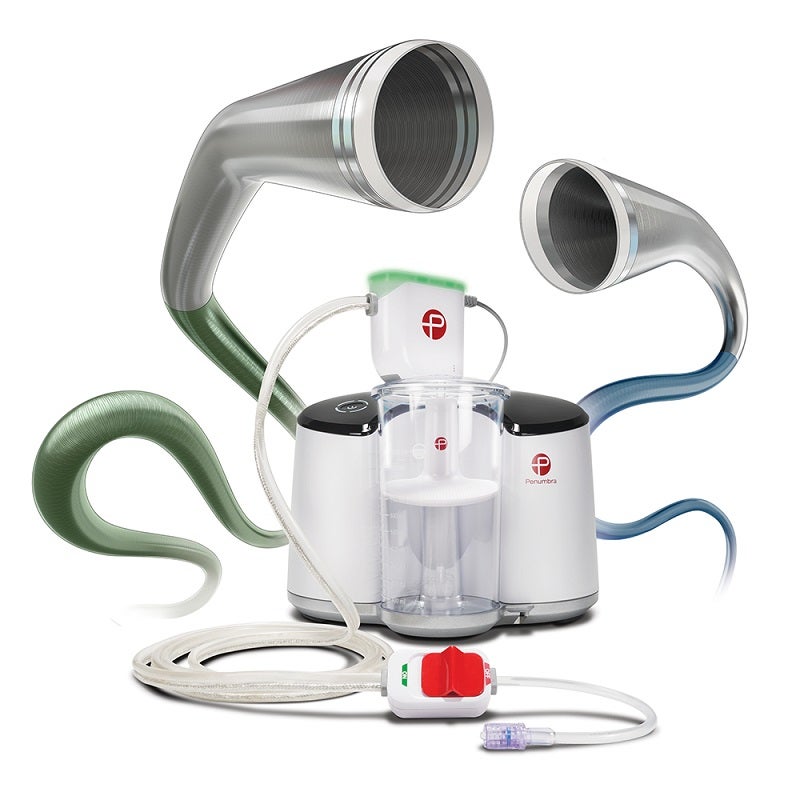
Penumbra and Japanese medical device manufacturer Asahi Intecc will collaborate to launch the Indigo Aspiration System in Japan following receipt of regulatory approval.
A minimally invasive device from Penumbra, Indigo can be used to remove emboli and thrombi from peripheral arterial and venous system vessels. It can also be used to treat pulmonary embolism.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Additionally, the device enables the restoration of blood flow in cases of venous thrombus and acute limb ischemia.
The latest generation offering combines the hypotube-based Indigo Aspiration Catheters with Lightning Intelligent Aspiration.
The Lightning Intelligent Aspiration is a computer-based clot detection technology that can distinguish between clots and blood.
It is intended to reduce blood loss as well as the use of clot-dissolving drugs, which can cause bleeding complication risks.
Penumbra CEO and president Adam Elsesser said: “By bringing together our newest innovations with Asahi’s leadership and expertise in the Japanese medical device market, we can provide physicians in Japan with technology that has been proven highly effective, helping patients return home quickly and in good health.
“This furthers our commitment to advance novel technologies to address unmet needs and improve patient care worldwide.”
Medico’s Hirata, a medical device trading company, will continue to distribute Penumbra’s devices for neurovascular thrombectomy as well as neurovascular and peripheral embolisation.
These devices also include the new RED reperfusion catheters, which are intended for use in the revascularisation of acute ischemic stroke patients.
With optimised trackability and aspiration efficiency, the new catheters are designed to help navigate the brain’s complex anatomy and provide efficient aspiration to remove blood clots in a wide range of stroke patients.





