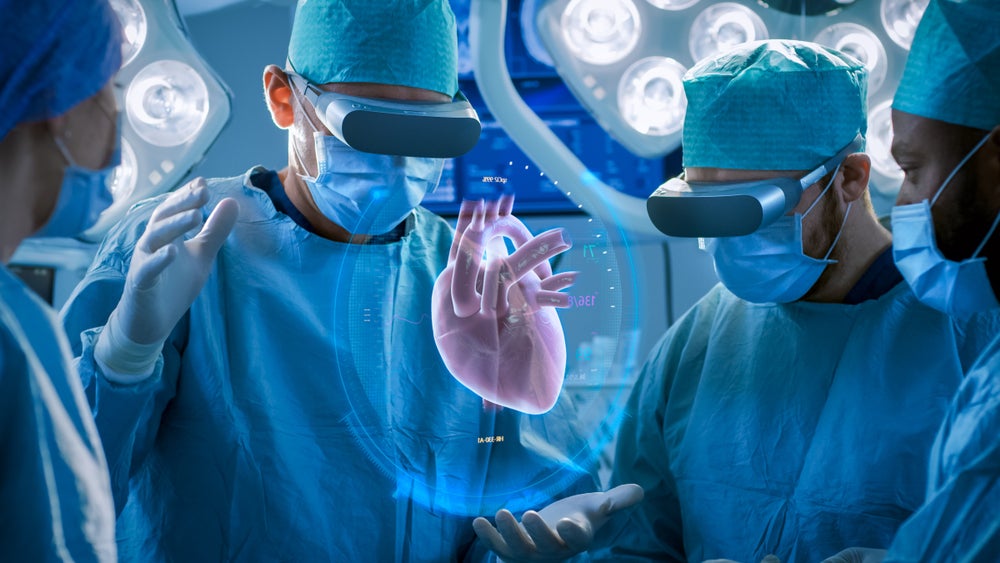
France-based Avatar Medical has joined the virtual reality (VR) wave sweeping through the healthcare sector, after the US Food and Drug Administration gave a 510(k) clearance for the company’s software.
Avatar Medical’s VR platform harnesses data from computerised tomography (CT) and magnetic resonance imaging (MRI) scans to generate a patient avatar – a 3D representation of the imaged body region.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The company says its software can help surgeons plan for procedures and be used during surgical procedures themselves for reference. Some of the clinical applications include breast surgery, vascular surgery, orthopaedic surgery, and surgical oncology.
The technology can also be used for education purposes.
According to Avatar Medical, more than 100 surgeons across 20 hospitals and universities have already used the software.
A report by GlobalData forecasts the global VR market to be worth $51bn by 2030 – the main drivers of VR in healthcare are surgical application and education use.
City University of New York School of Medicine director of surgical education Dr Elliott Goodman said: “Avatar Medical has the potential to revolutionise medical education but also to impact actual patient care. With real-time image rendering and lossless representations, we can now see pathology and anatomy in a way that has never been realised. This will greatly impact how we as surgeons prepare for our procedures.”
Avatar Medical CEO Xavier Wartelle said: “The FDA’s clearance is a major milestone in getting our VR solution in their hands to care for their patients. We expect European medical device certification for next year.”



