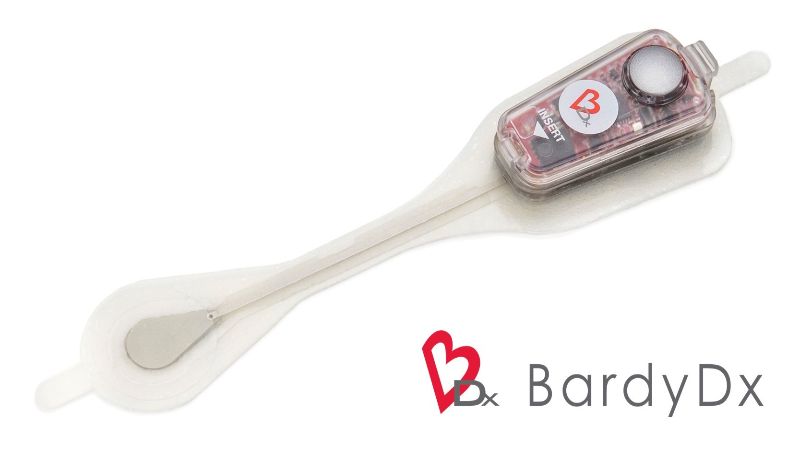
The US Food and Drug Administration (FDA) has granted 510(k) clearance to patient monitoring solutions firm Bardy Diagnostics’ 14-day variant of the Carnation Ambulatory Monitor (CAM) patch.
The CAM patch is a P-wave centric device that acts as an ambulatory cardiac patch monitor. Another use is for arrhythmia detection.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The patch is worn on the centre of the chest, directly over the heart, to ensure optimal capture of ECG signals.
The device is available in two-day and seven-day versions. The latest 14-day variant provides extended cardiac monitoring and potentially detect less-frequent arrhythmia.
Bardy Diagnostics founder and CEO Gust Bardy said: “Comfortable dermal ECG recordings that focus on the P-wave for up to 14 days carry the potential to minimise use of costly additional rhythm diagnostic tools.
“We are excited to receive clearance from the FDA to enable clinicians the option to monitor longer and anticipate incremental detection of accurate, medically actionable data to improve patient management.”
The CAM patch uses a technology that detects and records the P-wave, a small amplitude ECG signal from the atrium, providing an accurate diagnosis of arrhythmia and patient management.
Users can mail the device or take it to a physician’s office for analysis. Once the study has concluded, data transfer from the patch can occur within minutes.
In a comparator study, the CAM patch was found to detect 40% more arrhythmia, allowing more informed clinical decisions in 41% of patients when compared to the iRhythm Zio XT patch.
A separate study demonstrated significantly higher identifications of arrhythmia with Bardy’s device versus a standard Holter monitor. The cardiac patch was also able to detect arrhythmia that the Holter monitor missed or did not identify correctly.





