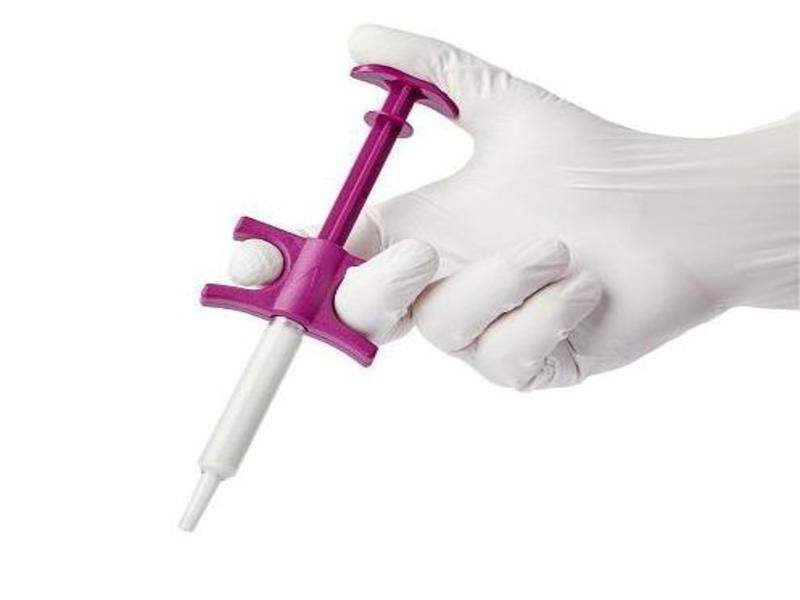
The US Food and Drug Administration (FDA) has cleared surgical products maker Baxter International’s new synthetic bone graft substitute Actifuse Flow for use in various orthopaedic surgical procedures.
Part of the company’s osteobiologics surgery range, Actifuse Flow is a prepackaged delivery syringe used to accelerate bone growth.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The graft substitute is intended to enable precise positioning into small bony voids or gaps in the skeletal system.
Actifuse Flow is based on Baxter’s Actifuse Bone Graft Substitute, which uses silicate-substituted technology to increase silicon levels for driving the formation of bone.
The new bone graft substitute does not require any prior mixing or preparation. It is said to retain its flowable consistency throughout the surgical procedure.
The syringe design enables the start and stop of delivery and can be used in less invasive surgical methods. This functionality also makes it ideal to fill small bone defects and complex geometries.
Furthermore, the graft substitute resorbs and is replaced by the patient’s own bone during the healing process. It is expected to be useful in pelvis, extremities and posterolateral spine orthopaedic surgeries.
Baxter Advanced Surgery business president Wil Boren said: “As part of our growing product portfolio, Actifuse Flow builds on the extensive clinical experience of our Actifuse Bone Graft Substitute.
“We strive to pioneer products that provide surgeons innovative and dependable tools to help enhance healing, improve outcomes and reduce the total cost of care.”
Actifuse Flow bone graft substitute, which is expected to be commercially launched over the coming months, will be available in 1.5ml, 3ml and 5ml sizes.
Other products under Baxter’s osteobiologics surgery portfolio include Actifuse ABX, Actifuse Shape, Actifuse MIS and Altapore.
All these products leverage silicate-substituted technology to accelerate bone growth. They are also available in a variety of configurations to address different requirements.





