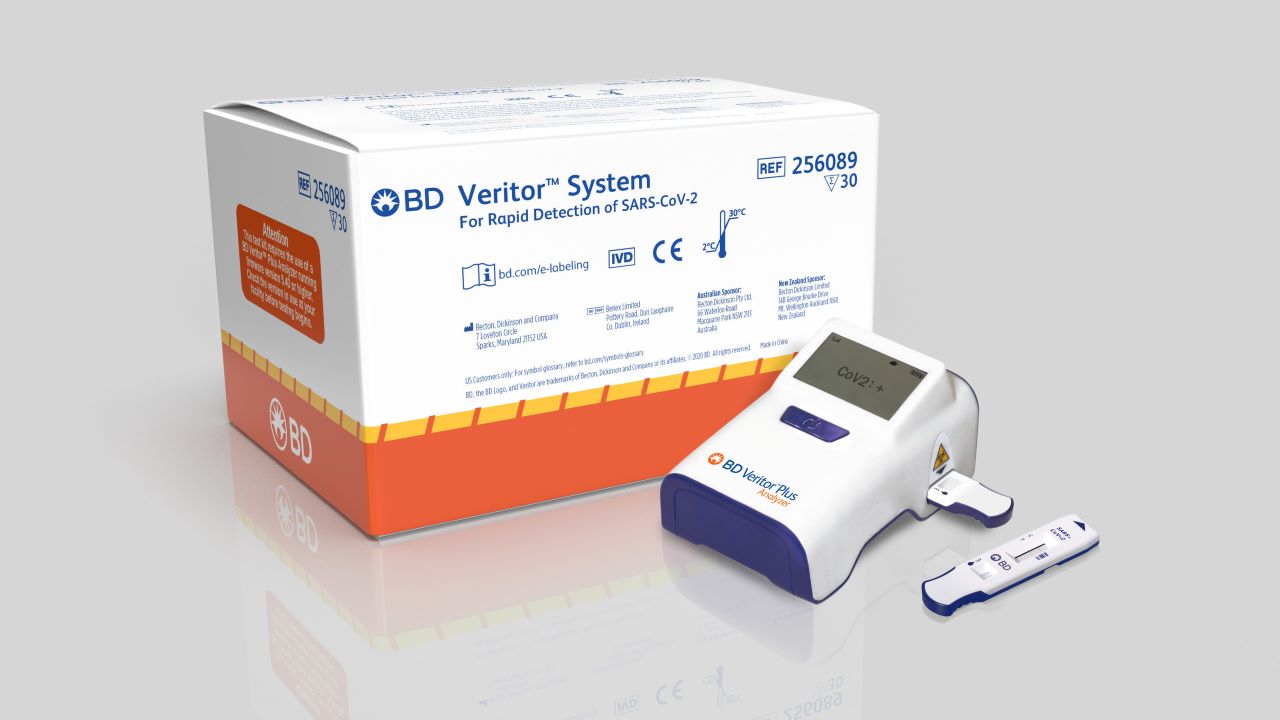
Becton, Dickinson and Company (BD) and ImageMover have signed an agreement to provide a companion mobile app for streamlining reporting of SARS-CoV-2 antigen testing results performed on the former’s Veritor Plus System in everyday settings.
The ImageMover app is available immediately for the users of the BD Veritor Plus System for Rapid Detection of SARS-CoV-2 Assay.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
It is authorised by the US Food and Drug Administration (FDA) for emergency use in detecting proteins from SARS-CoV-2.
In everyday testing locations such as schools and businesses, Covid-19 testing poses challenges in handling test subject demographics and results.
The app helps organisations to carry out point-of-care testing to effectively capture needed demographic details of people who are being tested, upload Covid-19 test results, report them to appropriate stakeholders, as well as automate reporting to federal and state agencies.
BD Veritor Plus System has in-built functionality to permit data sharing to electronic health records through the BD Synapsys Informatics platform. However, this functionality needs an information technology infrastructure common to diagnostic testing locations in healthcare facilities.
Adding the ImageMover app to the system offers seamless and secure data sharing for everyday locations without an established healthcare information technology platform.
BD Life Sciences president Dave Hickey said: “The ImageMover app helps resolve a major administrative reporting burden for ‘return to normal’ testing and further strengthens the advantages of using a digitally read test like the BD Veritor Plus System.
“From handheld portability to a simple workflow to a definitive digital readout of results that eliminates human subjectivity, and now, an automated reporting tool, the BD Veritor Plus System provides the simplicity needed for novice testers.”
Last week, BD acquired GSL Solutions, a US-based developer for smart medication devices, for an undisclosed amount.





