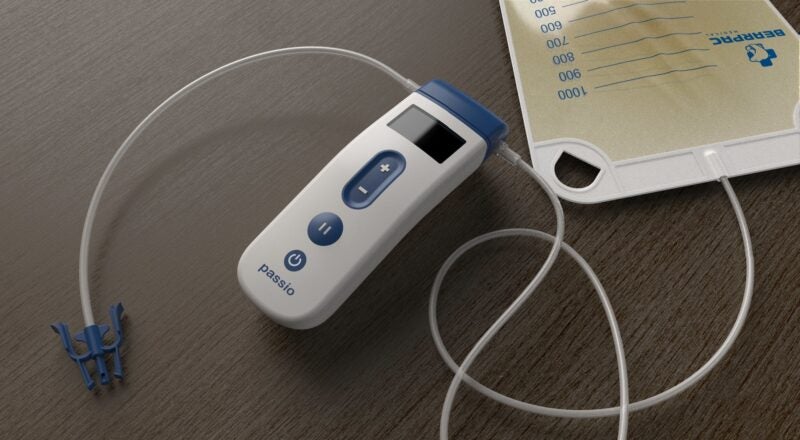
Bearpac Medical and APR Medtech have signed an exclusive distribution agreement for the sale of the Passio Pump Drainage System.
The system is intended for intermittent draining of recurring and symptomatic pleural effusions, as well as malignant ascites.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
APR Medtech technical director Michael Pichel said: “The Passio Pump Drainage System is an innovative, next-generation product and we are delighted to be partnering with Bearpac Medical to bring this technology to the NHS and UK private healthcare sector.”
The system comprises a Passio catheter, handheld control unit (pump) and disposable collection kit, which includes a redressing kit.
To perform fluid drainage, the disposable collection kit is used to connect the Passio pump to an implanted Passio catheter and the pump is activated to start the fluid evacuation into the collection bag.
The catheter is created specifically for use with the Passio collection system.
Compared to other devices currently available in the market, Passio offers flow control throughout the process at reduced vacuum pressures.
Bearpac Medical president Jay Zimmerman said: “This agreement with APR Medtech presents a significant market opportunity for our Passio Pump Drainage System, now with an expanded CE certification to include both pleural and peritoneal indications.”
The latest deal comes after the company obtained CE Mark for the usage of the Passio Pump Drainage System for draining recurrent and symptomatic pleural effusions and malignant ascites.
The certification enables the company to market the system in European Economic Area (EEA) member countries.




