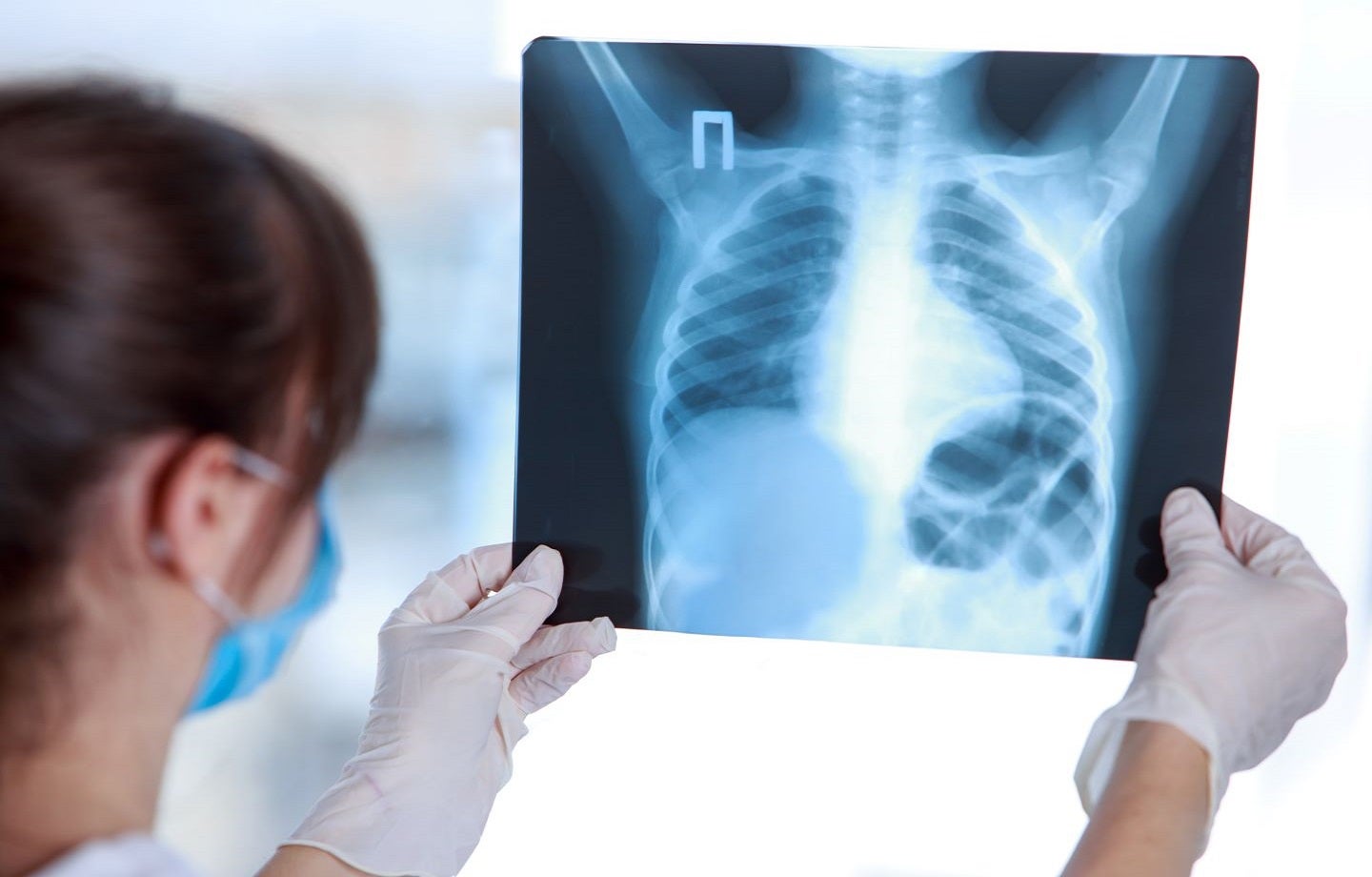
Bering has received the US Food and Drug Administration (FDA) 510(k) clearance for BraveCX, its chest X-ray triage solution powered by artificial intelligence (AI).
The company can now offer its AI solution commercially to healthcare institutions and medical professionals in the US.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The radiological computer-assisted triage and notification software is intended for analysing chest X-ray (CXR) images of adults (≥18 years old) to identify pre-specified suspected clinical findings.
BraveCX was developed to triage and prioritise emergencies like pleural effusion and pneumothorax immediately following the examination.
Physicians receive notifications of findings, offering a ‘second opinion’ and speeding up the diagnosis of urgent cases.
The AI-powered product was created using more than one million CXRs from various clinical settings and refined with input from over 50,000 CXRs labelled by board-certified radiologists.
BraveCX achieves specificity rates between 95% and 97% and ROC AUCs of 0.96 for pleural effusion and 0.98 for pneumothorax.
Bering CEO and founder Dr Ignat Drozdov said: “After over three years of research and collaboration with clinical teams, it’s so exciting to see BraveCX emerge as a state-of-the-art tool that has actually ‘listened to the end user’.
“FDA clearance means BraveCX prioritises patient safety, whilst still delivering the most advanced risk stratification algorithms where they are needed the most.”
By utilising a flexible deployment model for a cloud-based service—whether deployed directly on-premises or integrated with CXR hardware systems—the company intends to leverage both new and existing partnerships to swiftly launch the device into the market.
Established in 2010, Bering is a medical AI software company that focuses on developing clinical decision-support systems.



