Israeli biotechnology company BioProtect has raised $25m as part of its Series D equity financing from an undisclosed strategic investor and Peregrine Ventures.
The round, which was led by Almeda Ventures and Consensus Business Group chairman and investor Vincent Tchenguiz, had also seen the participation of KB Investments of South Korea and Triventures.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
BioProtect will use the proceeds from the round to support the ongoing multicenter Food and Drug Administration (FDA) clinical trial of its ProSpace balloon spacer and the expansion of its technology platform.
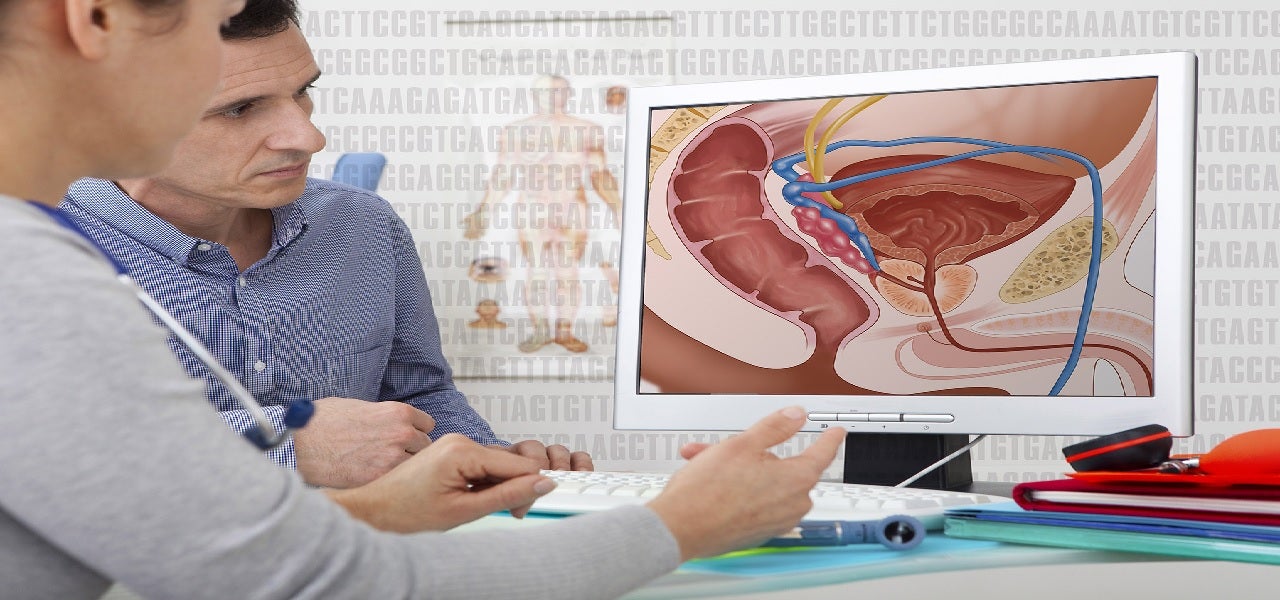
ProSpace balloon spacer is developed to protect prostate cancer patients who undergo radiation therapy.
Over 1.1 million men are diagnosed with prostate cancer every year globally and approximately 400,000 will undergo prostate radiotherapy. However, prostate radiation therapy has not always been recommended historically due to concerns over the risk of rectal toxicity, following radiotherapy.
The ProSpace balloon spacer helps to reduce radiation therapy risks by pushing the prostate away from the rectum.
Once injected, it provides physicians with a consistent gap of over 1.5cm, which is visible under trans rectal ultrasound and CT. The spacer is gradually absorbed after radiation therapy is completed.
BioProtect CEO Gil Rosen said: “This financing will allow the company to complete the pivotal study, gain FDA approval and accelerate the development of the next indication to our platform.”
“During the pandemic, and possibly even more so after the pandemic, there will be pressure on radiation oncology providers to shorten the treatment cycle without compromising safety and cancer control. First, in order to minimise the number of visits, and with them, pandemic risks to the patients, and later, once the pandemic subsides, the backlog of low-risk patients that delayed their treatment will be significant.
“We hope to provide an important enabling tool for these providers and patients.”
Meanwhile, in another major development in the field of oncology, Royal Philips has launched an advanced 3D imaging and navigation platform to accelerate lung cancer diagnosis and treatment.
Called Azurion Lung Edition, the system is designed for use in bronchoscopy procedures. It enables clinicians to perform both minimally invasive endobronchial biopsies and lesion ablation during the procedure.





