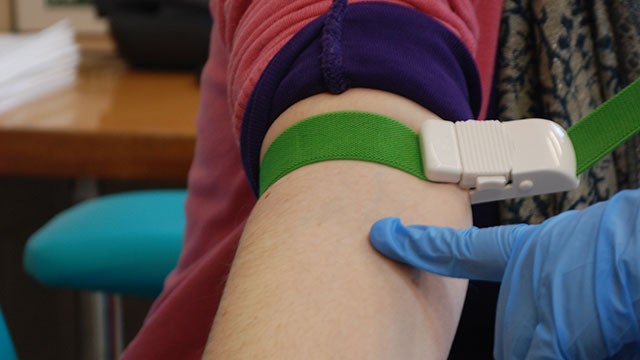
A noninvasive blood test has been developed by scientists in the US to predict a patient’s survival outlook after lymphoma diagnoses.
The test, created by scientists from Northwestern Medicine and the University of Chicago, identify if a patient with lymphoma will relapse after receiving treatment, predicting their survival time.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
On a molecular level, the test identifies the type of tumour the patient is suffering, indicating how long the patient might live or respond to treatment in a better way.
Northwestern University Feinberg School of Medicine associate professor of cancer epidemiology and prevention Wei Zhang said: “Lymphoma represents an increasingly important health issue in the United States, so developing novel approaches for precise care of these patients will improve patients’ clinical outcomes and life quality.”
Lymphoma’s incidence rate has nearly doubled in the US since the 1970s, with recent statistics suggesting that in 2016 alone, there were over 81,000 cases of lymphoma diagnosed in the country.
It is the latest discovery Northwestern scientists and collaborators used to identify patients’ liver cancer or diabetic complications in those with diabetes. Scientists are also testing it in other types of cancers, such as multiple myeloma and colon cancer.
Zhang said the goal is to test the technology on patients in a clinical trial, eventually bringing it into a real clinical setting.
“Ideally in the future, a patient could get their blood tested with this technology and check for a suite of different cancers,” Zhang said.
The technology has also been used by Zhang to analyse blood samples of over 3,000 people to accurately identify liver cancer in patients without mistakenly flagging those merely at risk.





