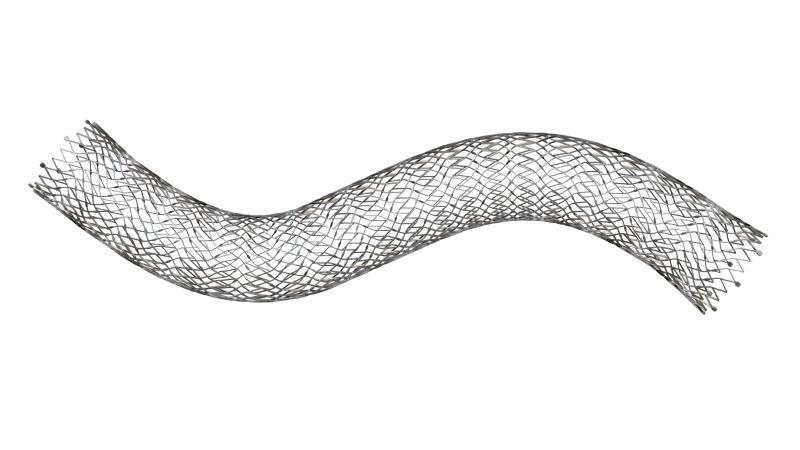
Boston Scientific has received approval from the US Food and Drug Administration (FDA) for the use of its Vici venous stent system to treat iliofemoral venous obstructive disease.
The disease is caused due to blockage of the blood flow through the veins located deep in the pelvic by a blood clot, or when the flow is compressed by anatomical anomalies.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Known to affect approximately 40% of people in the US, the disease could develop because of various conditions, including deep vein thrombosis (DVT), post-thrombotic syndrome (PTS) and compressive diseases such as May-Thurner syndrome.
Endovascular therapy for venous obstructive disease focuses on restoration of the normal flow of blood from the legs back to the heart.
Located deep in the pelvis, the iliofemoral veins are subjected to crushing forces from other anatomical structures such as the right common iliac artery.
The Vici venous stent system has been specifically developed to be uniformly strong and crush resistant. The device is capable of restoring blood flow by generating a cylindrical, patent vessel.
Boston Scientific Peripheral Interventions senior vice-president and president Jeff Mirviss said: “The FDA approval of the Vici venous stent system is the latest example of our commitment to building the most comprehensive portfolio of technologies specifically developed to meet the needs of physicians treating both chronic and acute venous disease.
“We are pleased to provide this differentiated stent system to US patients suffering from debilitating deep venous disease.”
The FDA approval was based on findings from the prospective, multi-centre, single-arm VIRTUS study that assessed Vici in a total of 170 patients.
During the study, the stent’s performance was determined based on pre-defined objective performance goals in patients with a clinically significant obstruction in the illiofemoral venous outflow tract.
The study met its primary safety and effectiveness endpoints.
Vici secured the European CE-Mark in 2013. The venous stent system was developed by Veniti, which was acquired by Boston Scientific in August last year.





