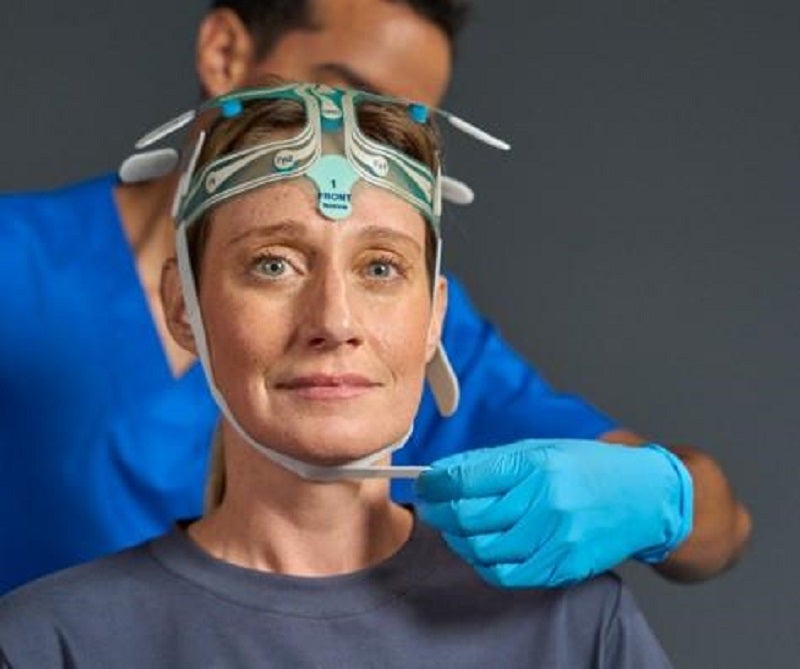
Medical device technology company Brain Scientific has introduced the improved and updated NeuroCap device.
The latest improvements to the device include a proprietary gel that offers better conductivity and enhanced performance adhesive pads.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
A base of modern blue PET material and a more conductive, precise ink formulation are also featured in the improvements.
The NeuroCap device has been developed to streamline electroencephalogram (EEG) preparation, testing and diagnosis.
It features a pre-gelled EEG headset with 22 electrodes and 19 active channels with fixed electrode placement according to the international 10-20 system.
The device allows healthcare providers to prepare a patient for an EEG exam quickly.
It can be used in a broad range of settings, including ambulances, athletic events, rural hospitals and private practices.
The company stated that the NeuroCap device has also received 510k clearance from the US Food and Drug Administration (FDA) for clinical use and is currently available in four sizes, including paediatric sizes.
Brain Scientific chairman and CEO Hassan Kotob said: “Our improved NeuroCap is a more comfortable, easy-to-use and efficient way to conduct EEG exams.
“The disposable NeuroCap, which is available for immediate shipping, allows clinicians to save prep time and see more patients, accelerating the timeline to results for critical EEG exams.”
Additionally, the company has signed a distribution partnership deal with medical equipment supplier D&D Medical.
Under the deal terms, D&D Medical will be responsible for selling Brain Scientific’s neurology devices in the US states of Indiana, West Virginia, Tennessee and Kentucky.
In August, Brain Scientific received CE Mark for its NeuroCap device.





