A two-year follow-up study of patients who received an Abbott HeartMate 3 cardiac pump has found that they had significantly lower rates of pump-related blood clots and strokes compared with heart-failure patients who had a commercial axial flow pump.
Results from the MOMENTUM 3 trial’s analysis at 24 months were presented in a Late Breaking Clinical Trial at the American College of Cardiology by Mandeep R. Mehra, MD, executive director of the Center for Advanced Heart Disease and medical director of the Heart & Vascular Center at Brigham and Women’s Hospital.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Mehra said: “This is a pivotal study in the field of advanced heart failure. Left ventricular assist devices have been in development for 40 years and there have been improvements in their technology but several challenges exist, including problems of blood clots forming in these devices, requiring device replacement. The field has been trying to engineer devices that could obviate some or all of these problems, and we report today on some important advances.”
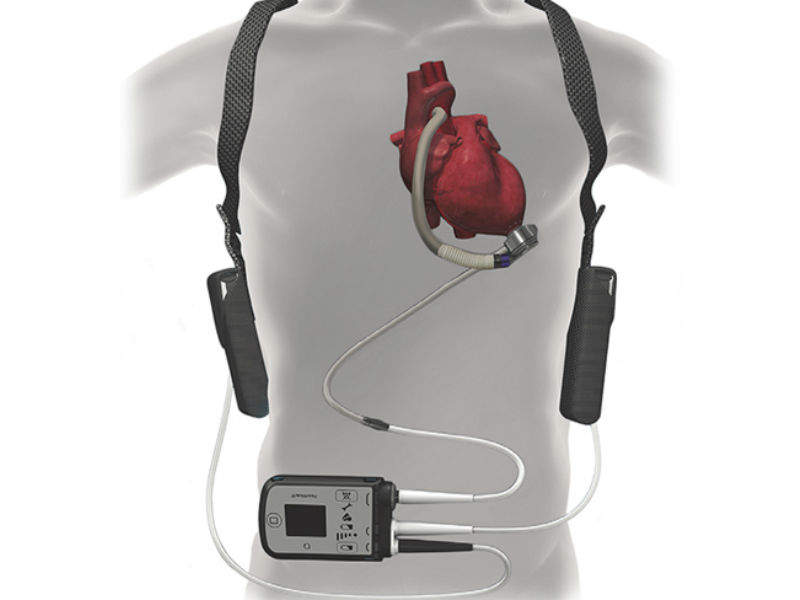
MOMENTUM 3, sponsored by Abbott, evaluated Abbott’s HeartMate 3 left ventricular assist system, which consists of a magnetically-levitated continuous centrifugal-flow circulatory pump, and compared it with the HeartMate II model. The trial evaluated how many participants, two years after receiving their device, had not suffered a disabling stroke or had an operation to replace or remove a malfunctioning device.
In the trial 366 patients were randomised to receive either the centrifugal flow pump or the axial flow pump. The researchers reported that 79.5% of patients on the centrifugal flow pump did not experience a disabling stroke or need a re-operation compared to 60.2% of patients on the axial pump.
Only three people who received the centrifugal-flow pump needed a re-operation compared to 30 who received the axial pump. No re-operations occurred due to blood clots in the centrifugal-flow pump. Deaths or disabling strokes were similar between the two groups, but overall, stroke rates were less frequent in the centrifugal-flow pump group. Bleeding and infection rates were no different between the two groups.
MOMENTUM 3 was launched in 2014 and was designed to dramatically reduce the overall timeline for clinical trials. All patients with refractory heart failure who needed a cardiac pump were eligible for the trial, regardless of whether the pump was intended as bridge to transplantation or destination therapy.
The HeartMate 3 includes several technological adaptations intended to reduce the risk of complications. Abbot claims that the fully magnetically levitated device runs like a bullet train – its rotor has no mechanical bearings in it and so it pushes the blood using only magnetism. It is designed to reduce stress, which is thought to cause blood clots to form in pumps.
In its next phase, MOMENTUM 3 will evaluate 1,028 patients at the two-year mark to further validate the current findings. Results of this are expected by the end of 2019.





