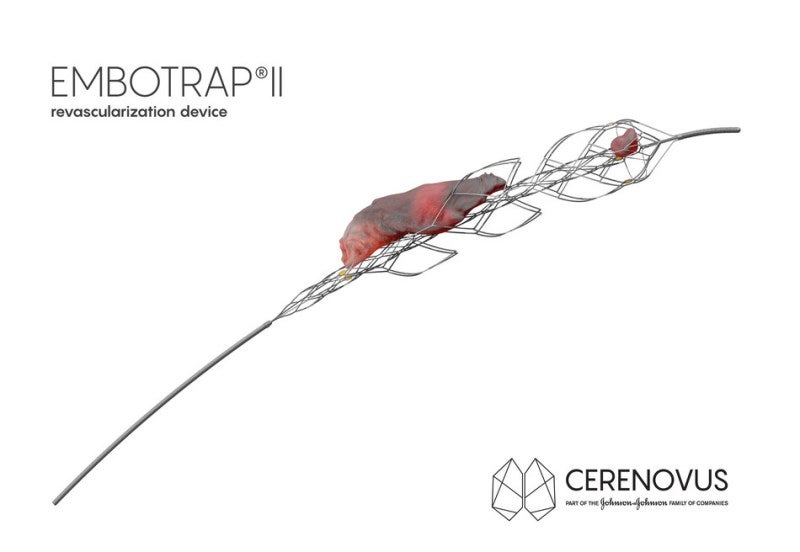
Cerenovus, a part of Johnson & Johnson Medical Devices Companies, has launched a new registry to gather and examine stroke-inducing blood clots removed from the brain with its EMBOTRAP II Revascularization Device.
The EMBOTRAP II Device is a next-generation stent retriever used to capture and remove life-threatening blood clots from the brain following an ischemic stroke.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Under the Excellent Registry, up to 1,000 ischemic stroke patients will be enrolled in about 50 clinical sites in the US and Europe.
Claimed to be the single largest global registry of its kind to date, investigators will collect evidence on all patients, preserve clots and then study to determine how different clot characteristics including size, composition and density are likely to affect clinical outcomes, patient comorbidities, and revascularisation rates.
Cerenovus president Daniella Cramp said: “This is a landmark registry study that will provide real-world data on the EMBOTRAP II Device while advancing the scientific community’s understanding of how variations in blood clots correlate with treatment and outcomes.
“We believe thoughtful and extensive research into the science of blood clots is the key to developing the most effective endovascular treatments and bringing its benefits to the greatest number of stroke patients.”
The EMBOTRAP II Device comes with a dual-layer design that enables doctors to maintain engagement and control a wide range of clots with just minimal compression while removing the clots.
This mechanical thrombectomy device, which has been available in Europe since 2016, secured US FDA clearance earlier this year.
It is indicated for use within eight hours of stroke symptom onset.
Earlier this month, the firm also received CE Mark approval of Geometric Clot Extractor (GCE) Revascularization Device, which is a stroke technology to retrieve various thrombus types, whether they are fibrin-rich thrombus (hard) or RBC-rich thrombus (soft).
Given the nature as well as the composition of some blood clots, around 20% or more of ischemic stroke cases are found to be resistant to standard procedures. To fill this gap, Cerenovus developed the GCE Revascularization Device.
Almost 80,000 people in the US suffer from strokes every year. It is considered to be a leading cause of disability, and the fifth leading cause of death. In the US, the economic burden of stroke is estimated at $34bn annually.
The company is currently undertaking a controlled evaluation study before launching the product commercially to analyse its clinical utility and understand its potential advantages over the currently available technologies.
Cramp added: “We are excited by both the clinical insights the EXCELLENT Registry will provide and the possibilities for GCE in helping to bring the benefits of mechanical thrombectomy to difficult to treat stroke patients.
“We have a deep commitment to meaningful innovation through the development of evidence-based solutions that give patients the best chance for a high quality of life after stroke.”





