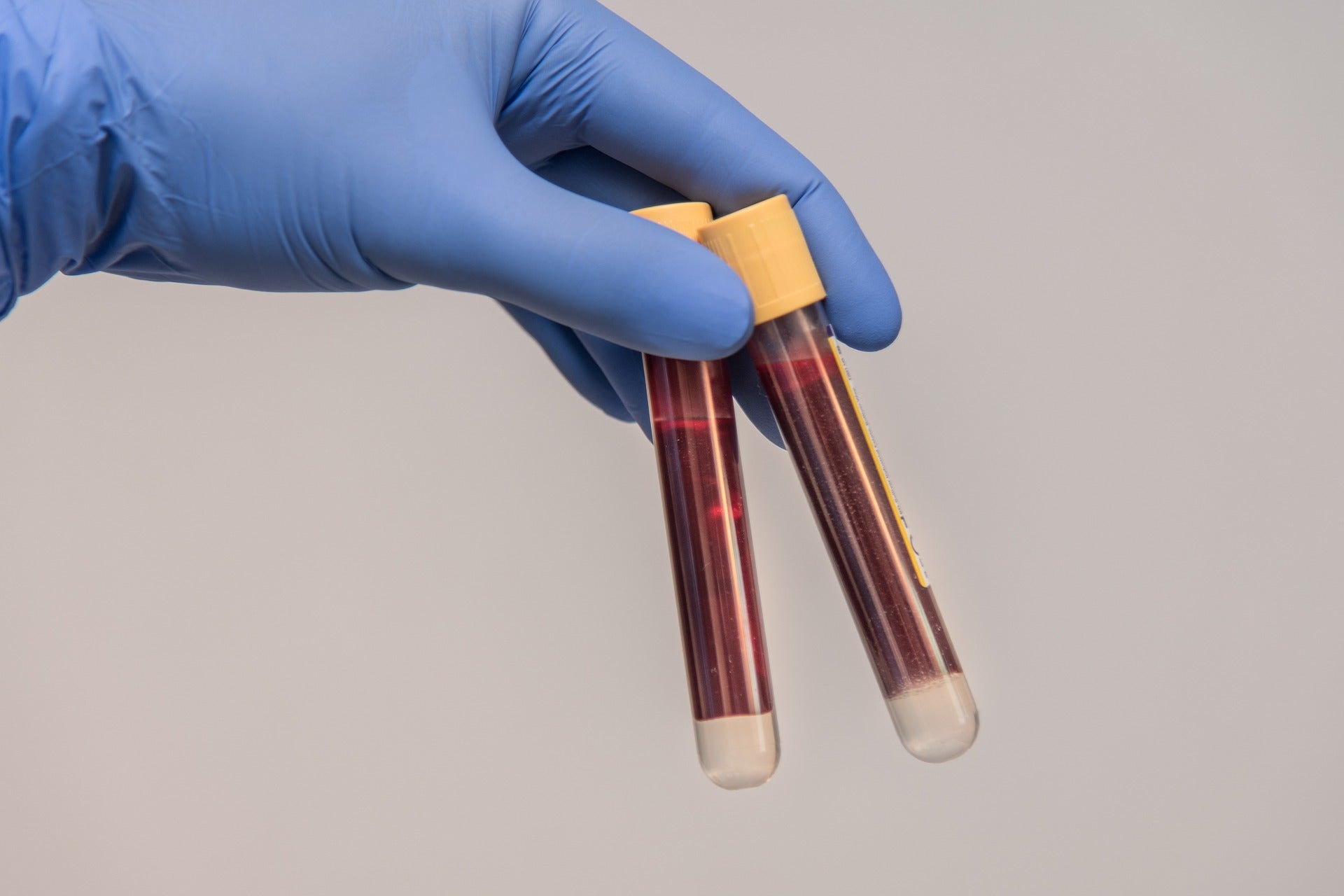
The New York State Department of Health Clinical Laboratory Evaluation Program (CLEP) has approved two non-invasive tests from Eurofins Viracor and Transplant Genomics that aid clinicians in analysing rejection status in kidney transplant recipients.
Eurofins Viracor and Transplant Genomics are part of the Eurofins Transplant Diagnostics group.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The Viracor Transplant Allograft Rejection Check (TRAC) donor-derived cell-free DNA (dd-cfDNA) test and TruGraf blood gene expression test aid health care providers in making decisions that help kidney transplant recipients achieve better long-term outcomes.
At present, invasive and expensive organ biopsy is the gold standard method of rejection diagnosis and surveillance, but it can cause additional complications for patients.
Viracor TRAC can be used to non-invasively identify rejection in renal transplant patients in the early stages.
The TruGraf test further aids clinicians by ruling out ‘silent’ subclinical acute rejection in kidney transplant recipients with stable renal function.
The simple, non-invasive blood test identifies an early variation in patients’ gene expression, as acute rejection activates the immune system before causing clinically observable symptoms.
With this early assessment of imminent rejection, clinicians can intervene and enhance kidney transplant outcomes.
Montefiore Medical Center kidney and pancreas transplant programme medical director Dr Enver Akalin said: “This is excellent news for kidney transplant patients in New York.”
With the latest CLEP approval, the TRAC and TruGraf tests are available to patients in all 50 US states.
Clinical labs carrying out clinical or forensic testing on specimens originating in New York State must have a New York State Department of Health clinical laboratory permit.
CLEP was established by the New York State Department of Health to guarantee test accuracy and reliability for its residents through strict evaluation standards.





