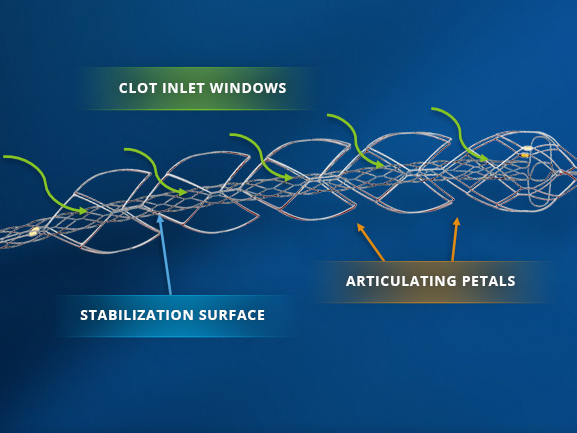
A tool for removing blood clots from the brain following an ischemic stroke, known as the EMBOTRAP Device, has demonstrated high success rates in restoring blood flow and patients achieving functional independence.
This is according to a new clinical trial published in Stroke, titled Analysis of Revascularization in Ischemic Stroke with EMBOTRAP Device (ARISE II).

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The EMBOTRAP Device, a next-generation stent retriever capable of retrieving a broad range of clot types, was developed by Neuravi. Cerenovus acquired Neuravi in April 2017.
During ARISE II, neurointerventional stroke physicians were able to restore blood flow in 80% of the patients treated with the EMBOTRAP Device within three passes (with a reperfusion grade of mTICI ≥2b). By the end of the procedure, vessels were opened in 93% of patients. Successful reperfusion was achieved with one pass in more than half the patients. At the 90-day follow-up, 67% were functionally independent. Overall, the study was considered a success.
Karolinska University Hospital neurosurgeon and neuroradiologist Dr Tommy Anderson said: “I believe that the ARISE II results demonstrate that EMBOTRAP can provide an effective treatment option for stroke patients with a large vessel occlusion. In particular, the high rates of complete revascularisation, with over half achieving successful reperfusion in a single pass.
“I have been using EMBOTRAP for a number of years, for the effectiveness and clinical outcomes, something I relate to the design of EMBOTRAP and how it functions in trapping the clot.”
ARISE II studied 228 patients in multiple clinics to assess the safety and effectiveness of the EMBOTRAP Device. Participants in the trial had large vessel occlusions and moderate to severe neurological deficits within eight hours of symptom onset.
Cerenovus head of clinical and regulatory ischemic stroke Mairsíl Claffey said: “We were honoured to work with an eminent team of international physicians committed to advancing stroke therapy.
“We are pleased that both the ARISE II results and the physician feedback during the study reflect positively on the EMBOTRAP Device design and the extensive clot research which forms its foundation. This research underpins our commitment to developing evidence-based stroke solutions to enhance patient care.”
Cerenovus submitted the ARISE II study data as part of its application to the US Food and Drug Administration (FDA) for marketing clearance of EMBOTRAP in the US. The device is already approved for use in Europe, where it has been used to treat more than 3,000 patients.





