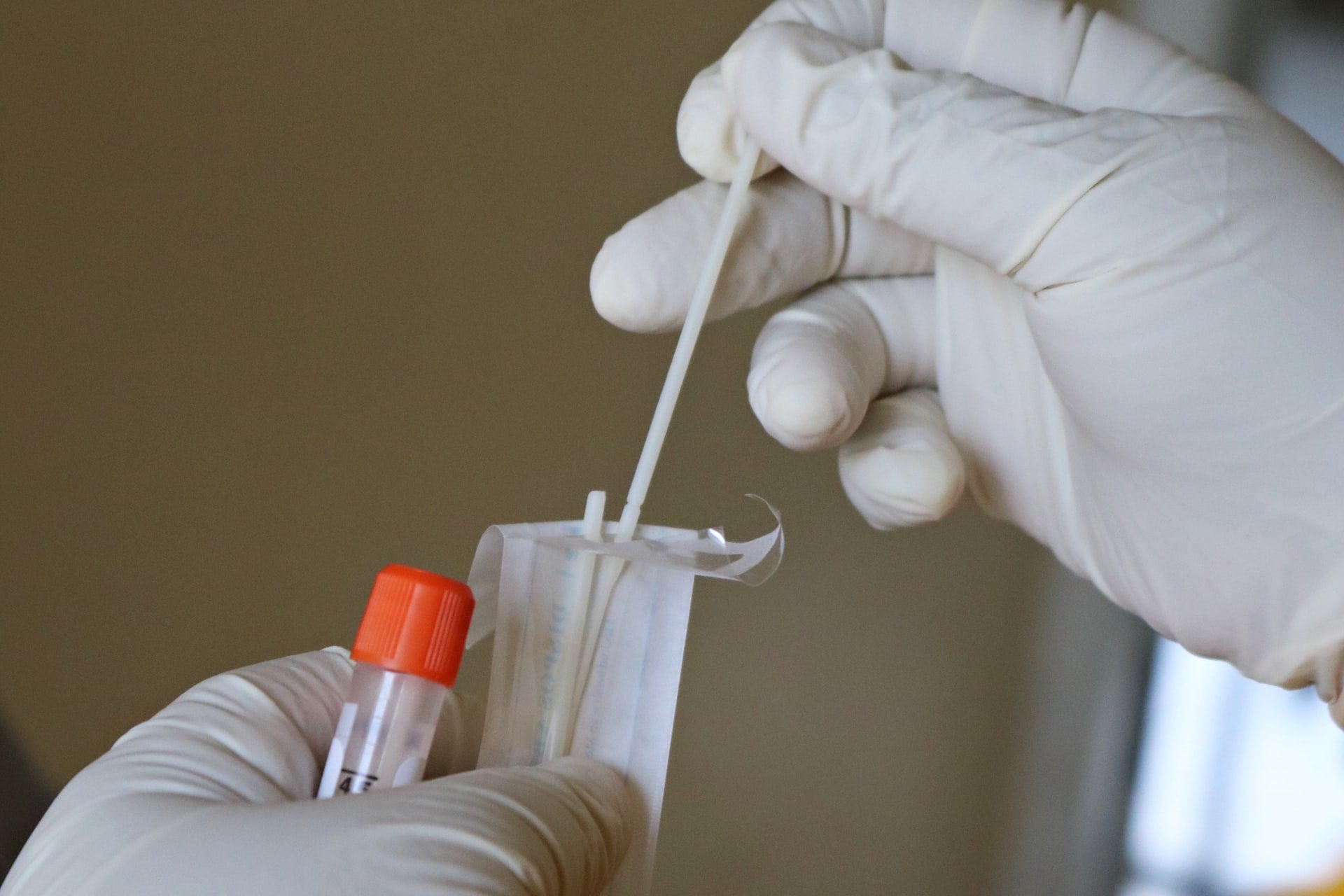
Co-Diagnostics has obtained CE mark to market its Logix Smart SARS-CoV-2 DS (Direct Saliva) as an in vitro diagnostic (IVD) for Covid-19 detection.
The test checks for the presence of the ribonucleic acid (RNA)-dependent RNA polymerase (RdRp) and E genes of SARS-CoV-2 in human saliva samples that are slightly processed.
It eradicates the need for RNA extraction from the samples, which is an expensive and time-consuming method used by many polymerase chain reaction (PCR) tests.
Co-Diagnostics noted that removing the extraction process could potentially boost throughput and reduce costs of testing for Covid-19 as well as other pathogens.
With the latest development, the new test will be available in all CE mark compliant markets and can also currently be purchased from the Co-Diagnostics Utah-based site in the US.
Co-Diagnostics CEO Dwight Egan said: “Our unique CoPrimer technology utilised in this test design received initial patent protection in 2018, which was further strengthened by an additional patent last year.
How well do you really know your competitors?
Access the most comprehensive Company Profiles on the market, powered by GlobalData. Save hours of research. Gain competitive edge.

Thank you!
Your download email will arrive shortly
Not ready to buy yet? Download a free sample
We are confident about the unique quality of our Company Profiles. However, we want you to make the most beneficial decision for your business, so we offer a free sample that you can download by submitting the below form
By GlobalData“In combination with this technology, direct saliva testing will play a key role in our ongoing centralised lab initiative, as well as our forthcoming at home and point of care testing strategies.”
The company noted that the CE marking for the Logix Smart DS CE-IVD Test establishes that it meets the Essential Requirements of the European Community’s In-Vitro Diagnostic Medical Device Directive, which allows the test to be exported and marketed as an IVD directly.
A molecular diagnostics company, Co-Diagnostics develops, produces and markets diagnostics technologies, which are used in tests for identification and/or analysis of nucleic acid molecules.
In March, the company developed a new point-of-care, at-home PCR testing platform to detect Covid-19 and other diseases using CoPrimer technology.
It uses the company’s DS extraction-free process and Logix Smart test reagents, which are pre-packaged in saliva receptacle tubes.



