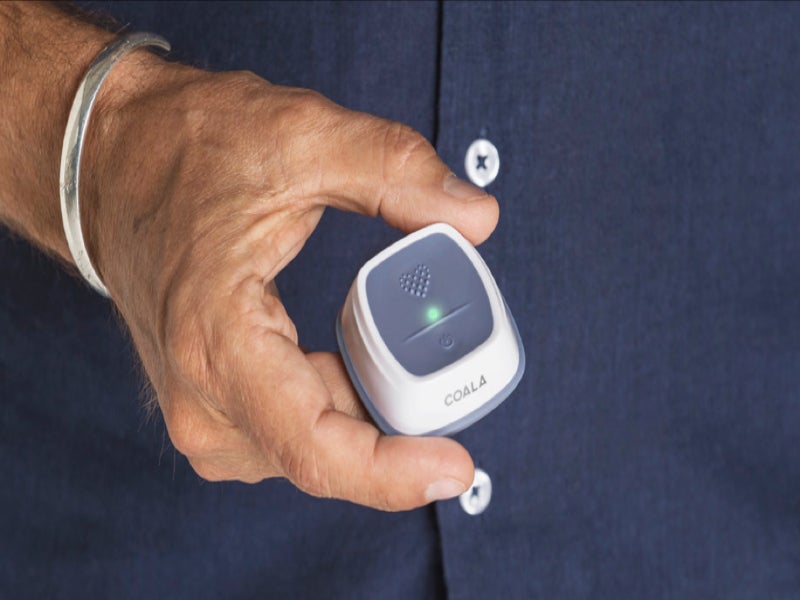
Swedish digital health company Coala Life has expanded the indication of its heart monitor for use in Covid-19 respiratory diagnostic exams.
The US Food and Drug Administration (FDA)-cleared Coala Heart Monitor can now be used to monitor heart and record lung sounds of the home quarantined patients in one minute. In addition, it enables physicians to remotely read the analysed data and view a patient’s electrocardiogram (ECG).

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
This wireless device can be activated from the patient’s homes, unlike other medical devices, which require hospital-based enrollment.
Coala Life founder and president Philip Siberg said: “The Coala device was developed and validated in Sweden based on over ten years of research. It truly enables telemedicine with the ability to monitor respiratory sounds and cardiac-related conditions in the safety of patient’s homes and help eliminate the need for patient office visits.”
Introduced in Scandinavia in 2017 for cardiac patients, Coala Heart Monitor has recently secured the US FDA approval.
The smartphone-powered device is integrated with a high-performance wireless stethoscope and a synchronous high-resolution 2-lead ECG. It enables real-time remote diagnostics of arrhythmias, forms of congenital heart disease and certain respiratory disorders in patients.
Following FDA’s new enforcement policy to support patient monitoring during the Covid-19 pandemic, Coala’s respiratory monitoring solution will be available for purchase by prescription for existing and new users in the US and selected EU markets.
The company said that devices like the Coala Heart Monitor are in high demand in Europe due to the fast-spreading Covid-19 outbreak.
It allows home-based management of high-risk patients with coronary and pulmonary diseases.





