Medical imaging IT and cybersecurity company Sectra has received US Food and Drug Administration’s (FDA) 510(k) clearance for its Digital Pathology Module.
The module is part of the company’s overall pathology solution. The clearance allows clinicians to use it with Leica Biosystems’ AT2 DX scanner.
Furthermore, healthcare providers in the US can use the digital pathology solution for primary diagnostics. It is already in use in the country for research and tumour boards.
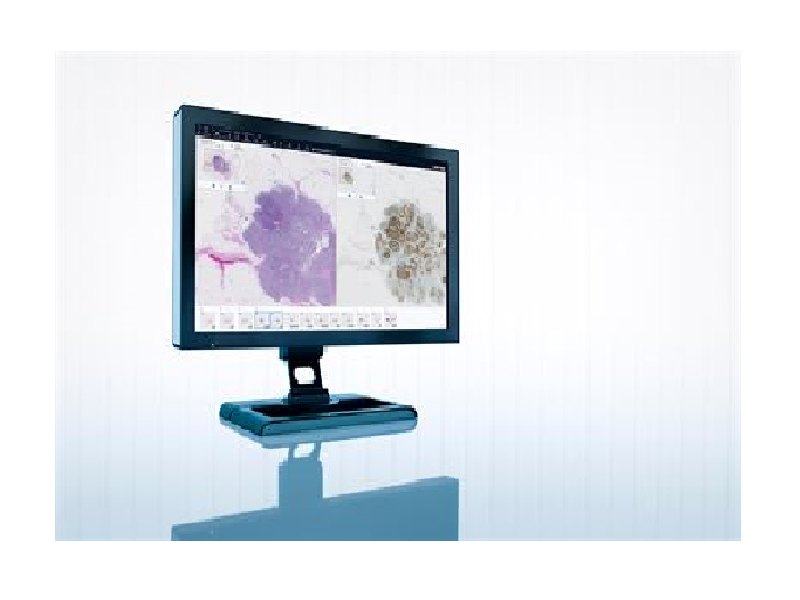
Pathologists can use the module for instant access to existing and historical images and related data, using digital images of tissue samples instead of physical glass slides.
In addition, the solution will allow efficient second opinions, remote reading, specialist consultations and integrated diagnostics.
Sectra president Mikael Anden said: “I am excited that with the FDA approval, we are now able to offer US healthcare providers a digital pathology, offering with a proven track record of use for full-scale primary diagnostics and large volumes.
How well do you really know your competitors?
Access the most comprehensive Company Profiles on the market, powered by GlobalData. Save hours of research. Gain competitive edge.

Thank you!
Your download email will arrive shortly
Not ready to buy yet? Download a free sample
We are confident about the unique quality of our Company Profiles. However, we want you to make the most beneficial decision for your business, so we offer a free sample that you can download by submitting the below form
By GlobalData“Our solution makes it possible for pathologists across the country to continue their important work from home without the need for a microscope. We look forward to providing uninterrupted pathology reading in these challenging times.”
The module enables clinicians to use a single solution for their medical imaging requirements across all specialities and allows integrated diagnostics.
In addition, it improves efficiency and patient care by providing deeper cooperation and the ability to access other departmental images and information.
Sectra CEO and president Dr Torbjörn Kronander said: “Pathology is the next huge digitisation of healthcare. The use of glass slides limits growth in today’s increasingly digital environment, especially in consolidated health care systems where potential synergies cannot be reached unless pathology is digitised.
“Our solution opens up new opportunities in the US where we can point to our successes in Europe in large production centres.”