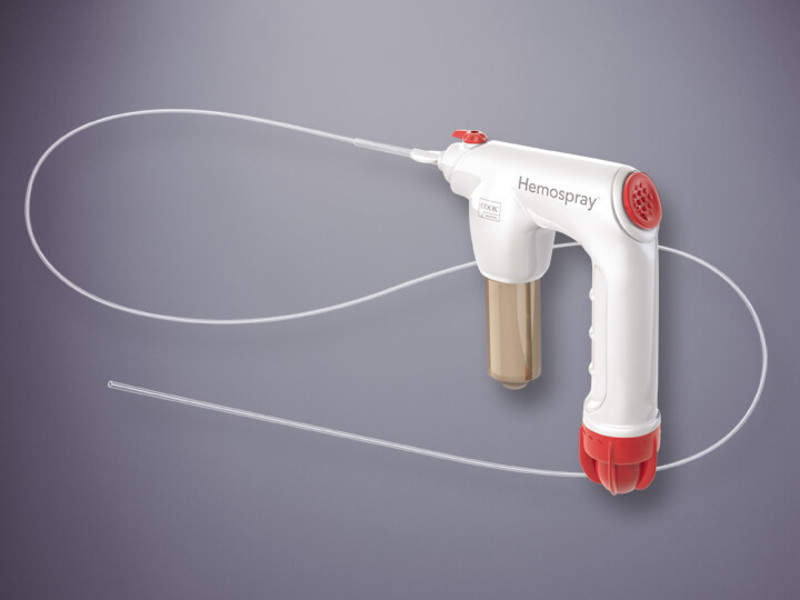
Cook Medical has obtained clearance from the US Food and Drug Administration (FDA) to market its Hemospray device for the treatment of some types of non-variceal gastrointestinal (GI) tract bleeding.
Indicated to control both upper and lower GI bleeding, the device has been designed as an aerosolised spray that delivers a mineral blend (inorganic powder) to achieve haemostasis at the target site.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
At the bleeding site, the powder absorbs water from the blood and forms a gel with a cohesive and adhesive nature that leads to the generation of a stable mechanical barrier over the bleeding site.
The single-use, non-thermal device can be applied during an endoscopic procedure and is said to cover large areas, including big ulcers or tumours.
FDA Center for Devices and Radiological Health surgical devices division director Binita Ashar said: “The device provides an additional, non-surgical option for treating upper and lower GI bleeding in certain patients, and may help reduce the risk of death from a GI bleed for many patients.”
The FDA clearance is based on findings from clinical studies involving a total of 228 subjects with upper and lower GI bleeding. This was further supported by real world evidence from medical literature reports of additional 522 patients.
Data indicated that Hemospray stopped GI bleeding in 95% of patients within five minutes of usage, while re-bleeding within 72 hours, up to 30 days after usage, was found in 20% of the subjects.
Meanwhile, around 1% of the participants are reported to have demonstrated the serious side effect of bowel perforation.



