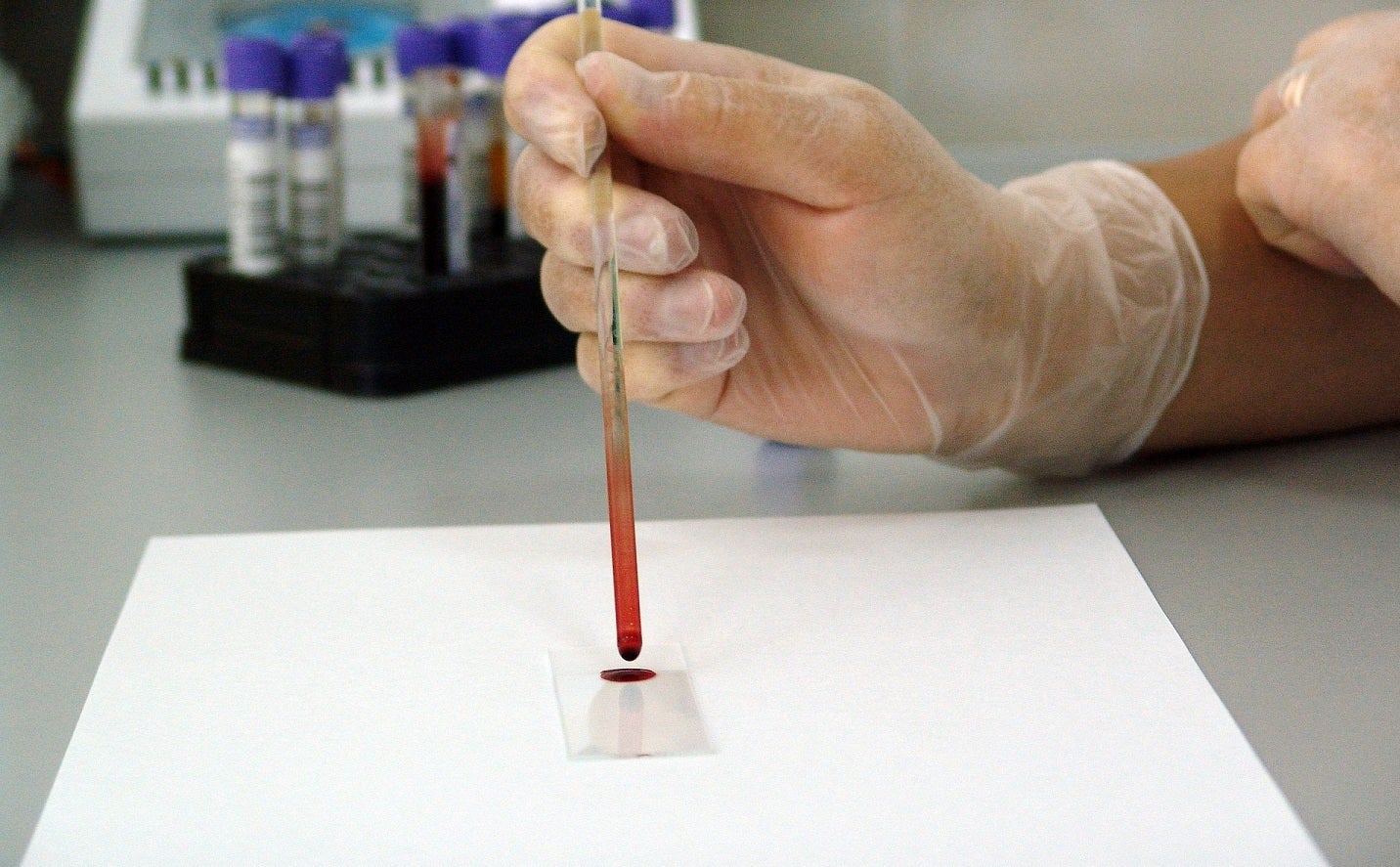
Daxor Corporation is seeking approval from the US Food and Drug Administration (FDA) for its next-generation blood volume analyser, Daxor BVA.
Daxor BVA is designed for rapid, simple, and bedside patient care.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The company submitted to the FDA through the 510(k)/CLIA-waiver dual pathway, with potential clearance for the analyser anticipated in the first half of this year.
Daxor CEO and president Michael Feldschuh said: “Today marks a long-anticipated and momentous milestone in our continued commitment to our customers and patients, and to achieving our vision of optimal blood volume for all.
“Funded under contract by the US Department of Defense, our next-generation analyser will deliver a level of speed, access, and accuracy to blood volume management that can significantly improve care and outcomes in medicine.”
The Daxor BVA system is designed to quantify the intravascular blood, red blood cells, and plasma volumes directly. This provides healthcare professionals with critical data to optimise fluid management for patients.
The new device boasts a threefold increase in speed, simplified operation, portability, and the ability to perform analyses at the patient’s bedside compared to the existing Daxor BVA-100 system.
These enhancements are expected to facilitate the integration of this advanced diagnostic tool into clinical routines, the company said.
Currently, the Daxor BVA-100 system is being used daily in major centres across the US. More than 65,000 tests have been performed to evaluate blood volume using this device.
Said to be the first diagnostic blood test cleared by the FDA, the BVA-100 provides safe, accurate, objective quantification of blood volume status and composition for use in a broad range of medical and surgical conditions.





