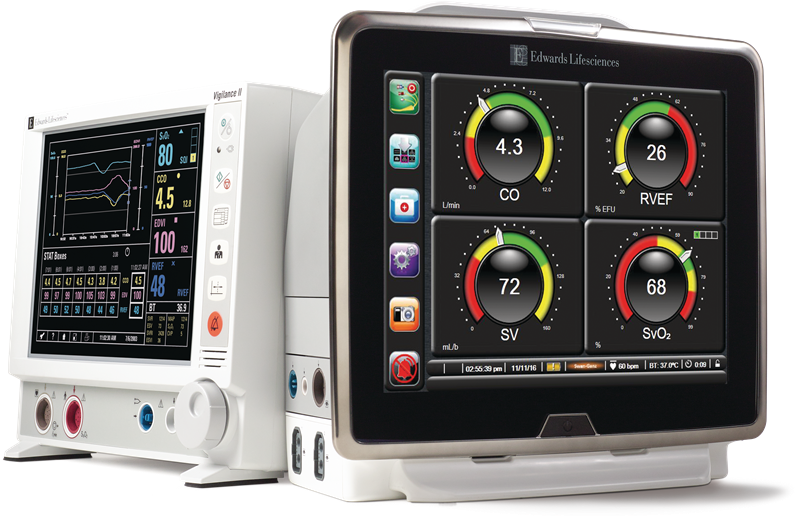
Edwards Lifesciences has signed a definitive agreement to acquire medical technology company CAS Medical Systems (CASMED) in an all-cash transaction valued at around $100m, or $2.45 per share.
CASMED focuses on the non-invasive monitoring of tissue oxygenation in the brain.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
It provides hospitals, emergency medical services and original equipment manufacturers (OEMs) with FORE-SIGHT Cerebral Oximeters to detect tissue hypoxia.
The oximetry products are said to accurately and non-invasively measure tissue oxygenation in the brain.
Edwards Lifesciences expects the FORE-SIGHT technology to complement its HemoSphere hemodynamic monitoring platform.
Edwards Lifesciences critical care corporate vice-president Katie Szyman said: “Cerebral oximetry technology provides an important indicator of oxygen levels in the brain, which can enhance clinician decision-making.
“We believe the incorporation of CASMED’s FORE-SIGHT technology into Edwards’ leading hemodynamic monitoring platform, along with Edwards’ predictive analytics capability, will strengthen Edwards’ leadership in smart monitoring technologies by providing physicians with a more comprehensive status of their surgical and critically ill patients.”
The company, in alliance with CASMED, developed a smart cable and software module that facilitates the connection between the FORE-SIGHT sensor and the HemoSphere monitoring platform.
While this technology has obtained the European CE-Mark, it is yet to receive 510(k) clearance from the US Food and Drug Administration (FDA).
CASMED president and CEO Thomas Patton said: “We are pleased with this opportunity to bring together CASME’s expertise in brain and tissue oxygenation monitoring with Edwards’ long history of leadership in hemodynamic monitoring.
“We are confident that the combination of these technologies will result in an important measurement tool for physicians that can empower them to improve care.”
Subject to customary closing conditions, the acquisition is set to be completed in the second quarter of this year.





