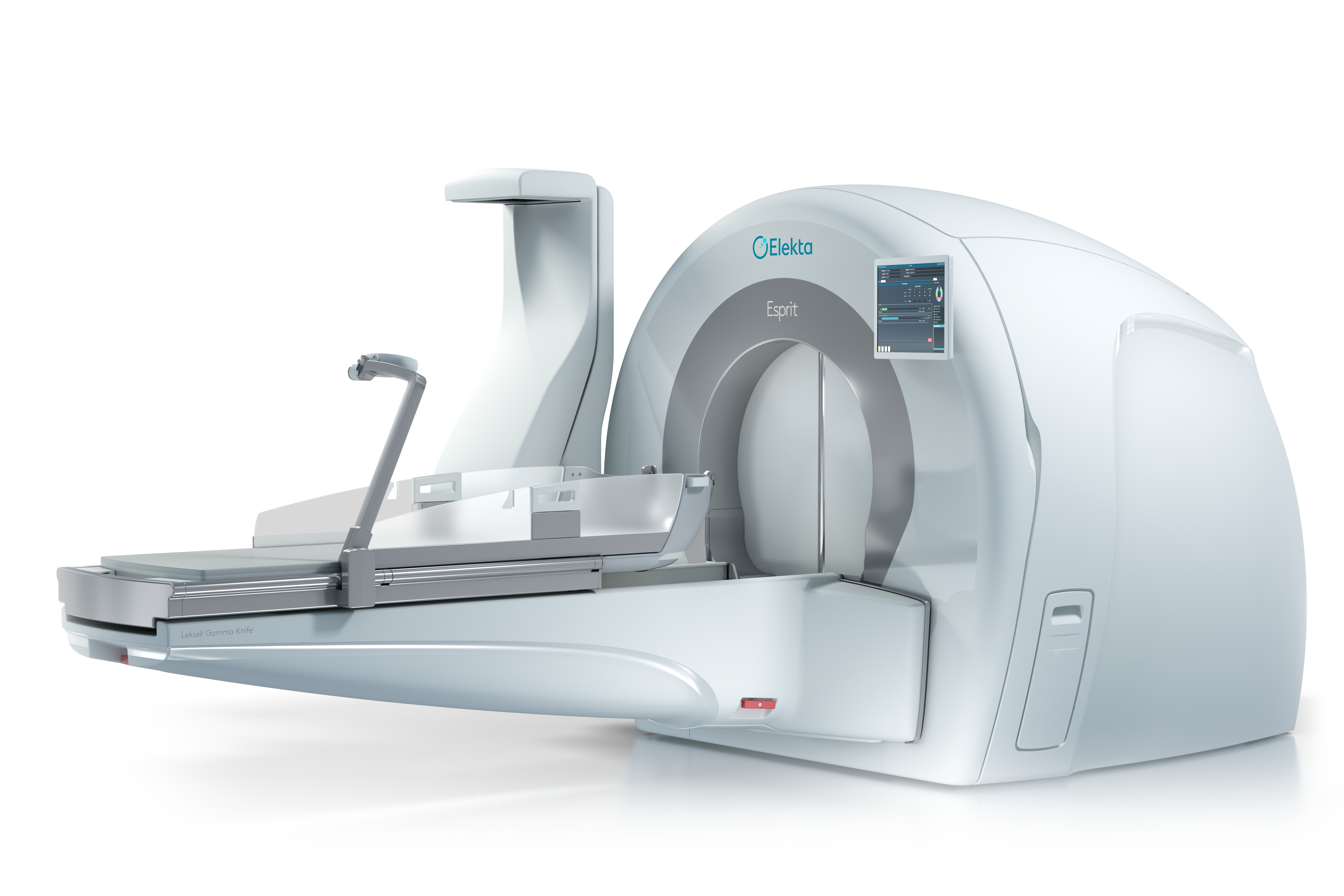
Swedish medical equipment firm Elekta has received 510(k) clearance for its new Elekta Esprit radiosurgery system from the US Food and Drug Administration (FDA).
A new Leksell Gamma Knife radiosurgery platform, Esprit serves as an advanced alternative to conventional radiotherapy and open surgery.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The new Leksell Gamma Knife helps clinicians target challenging intracranial tumours and lesions while protecting the motor, sensory and neurocognitive functions of a patient’s brain.
The approval will make Esprit accessible to clinicians across the US as well as in countries that recognise FDA-approved systems.
Elekta Neuroscience Solutions president Verena Schiller said: “It’s very inspiring to hear how patients treated with Gamma Knife are able to maintain their quality of life and go back to what they love doing after radiosurgery.
“Thanks to the very low dose delivered outside the target, it’s able to protect surrounding healthy tissue. Esprit takes Gamma Knife to the next level with improvements in both the patient and user experience.”
Elekta Esprit, unveiled in May, provides accurate visualisation to clinicians through frameless and frame-based workflows.
The platform also offers remote accessibility and collaboration tools to help clinicians treat challenging targets and maintain a personalised approach to radiosurgery.
Esprit ensures safe treatment for multiple metastases, arteriovenous malformations, trigeminal neuralgia and vestibular schwannoma.
In the same month as the Esprit launch, Elekta and the Netherlands Cancer Institute reached a ten-year strategic partnership and research and development alliance to develop the next generation of radiation therapy solutions and introduce new treatments to patients.
The parties agreed to expedite improvements in Elekta’s Unity and linac solutions by combining their expertise, resources and knowledge.



