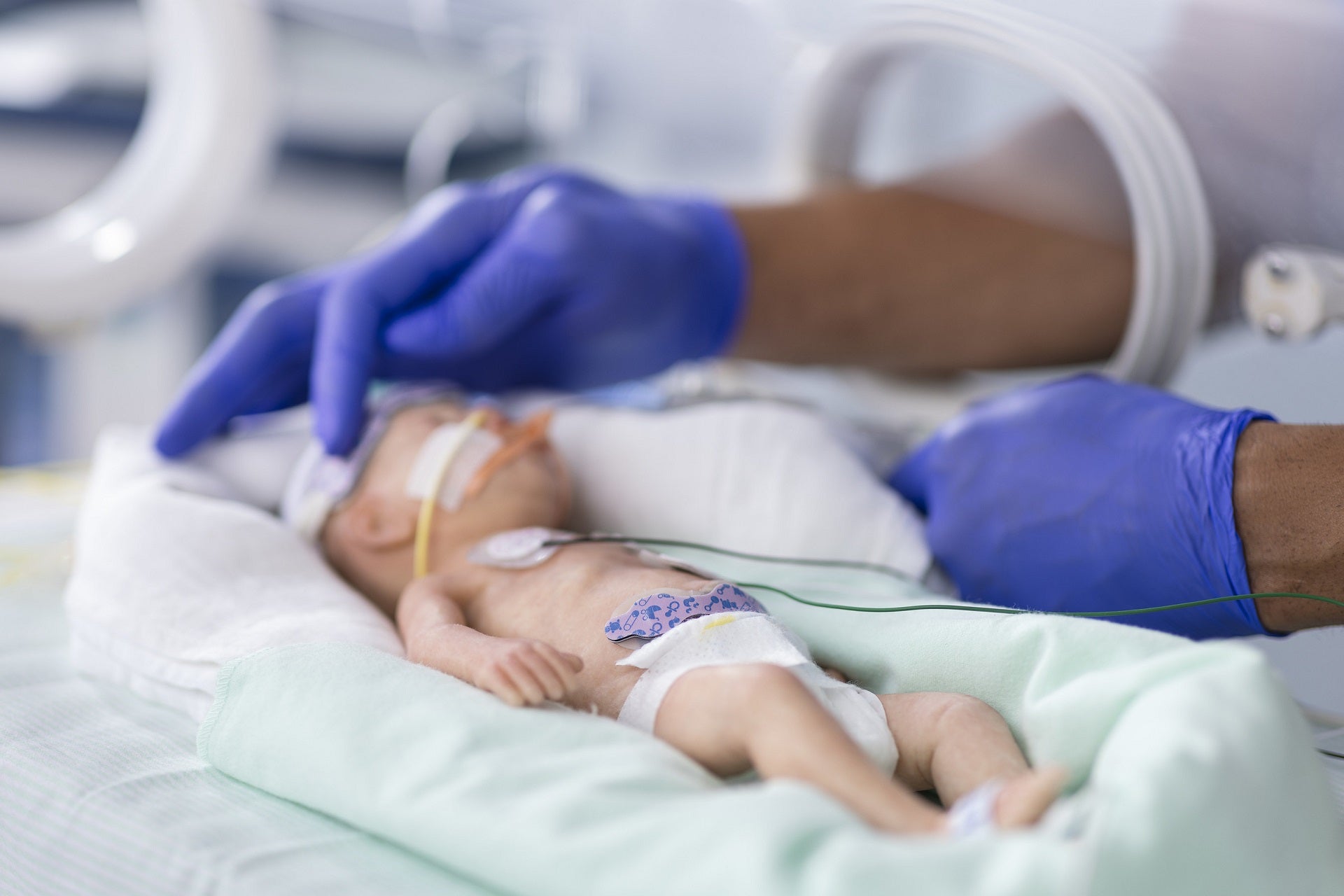
Medtronic has secured 510(k) clearance from the US Food and Drug Administration (FDA) for its INVOS 7100 cerebral/somatic oximetry system for paediatric indications.
The clearance will allow the use of the system for children from birth to 18 years.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The INVOS system is designed to provide key signals that will support time-critical decisions taken by paediatric clinicians.
It indicates problems related to ventilation, hemodynamic management and resuscitation for neonates, children, premature infants and other patients treated by paediatric clinicians.
Additionally, the system provides continuous measurement of cerebral oxygen saturation and reliable indication of changes in cerebral perfusion.
It also measures changes in regional oxygen saturation (rSO₂) levels of blood in up to four site-specific areas, which can be chosen by clinicians.
This provides better monitoring of organ-specific oxygen levels and allows the identification of warning signs for neonates and children in advance.
Medtronic patient monitoring business president Frank Chan said: “Timing is critical for vulnerable pediatric patients, and the INVOS 7100 system can alert clinicians to changes in patient condition before traditional monitored parameters even react.
“The INVOS 7100 system can help clinicians decide if intervention is necessary – a core component in successful outcomes. And, our technology consistently enables users to determine if they need to intervene sooner.”
INVOS 7100 measures tissue perfusion and oxygenation in real-time and alerts clinicians regarding changes in perfusion prior to other vital sign measurements.
This data may indicate a critical situation and can provide clinicians with the crucial time required to treat young patients and newborns.
The new system is designed with unique algorithms that measure acute alterations in rSO₂, oxygen metabolism and hemodynamics.
Medtronic will make the INVOS 7100 system with paediatric indications available across the world for commercial use next year.
Last month, Medtronic’s PillCam Small Bowel 3 (SB3) @HOME endoscopy procedure secured 510(k) clearance from the US FDA.





