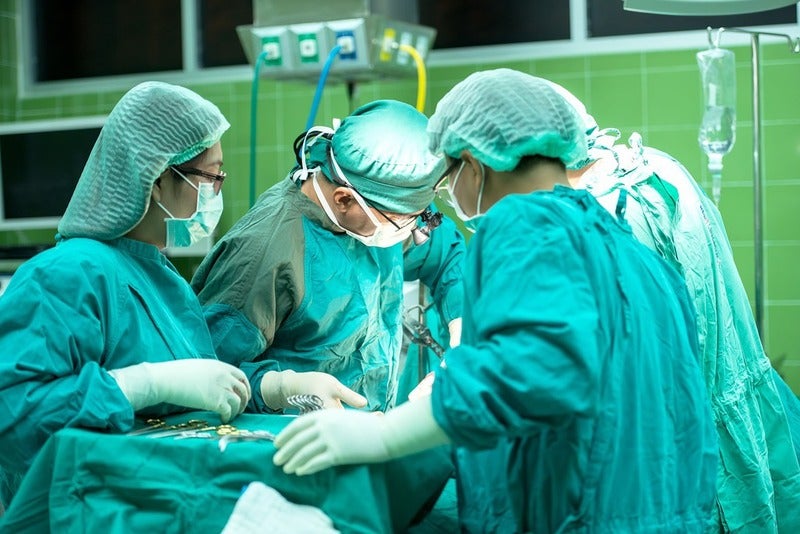
The US Food and Drug Administration (FDA) has alerted healthcare providers that a large number of medical device reports (MDR) submitted in the last seven years were related to surgical staplers and implantable staples.
In its letter, the FDA provided recommendations to help mitigate risks associated with the use of these devices.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
This move was made after the regulatory agency found it received more than 41,000 individual MDRs for surgical staplers and staples between 1 January 2011 and 31 March 2018.
The reports included 366 deaths, more than 9,000 serious injuries and around 32,000 malfunctions. The FDA is continuing to assess the MDRs.
The FDA Center for Devices and Radiological Health chief medical officer William Maisel said: “The agency’s analysis of adverse events associated with surgical staplers and implantable staples is ongoing, but we know these devices provide important benefits for patients undergoing surgery, so it’s important for us to continue to educate providers about the devices’ safety and risk.
“We are asking providers to be aware of the new information and implement the recommendations we’re outlining today to help improve the safe use of these devices.”

US Tariffs are shifting - will you react or anticipate?
Don’t let policy changes catch you off guard. Stay proactive with real-time data and expert analysis.
By GlobalDataSurgical staplers and staples may be indicated for use in a variety of surgical applications to remove part of an organ, cut through tissues, or create connections between structures.
The FDA will issue draft guidance with labelling recommendations for manufacturers and hold a public meeting to discuss if the current approach for manufacturers to market surgical staplers is appropriate.





