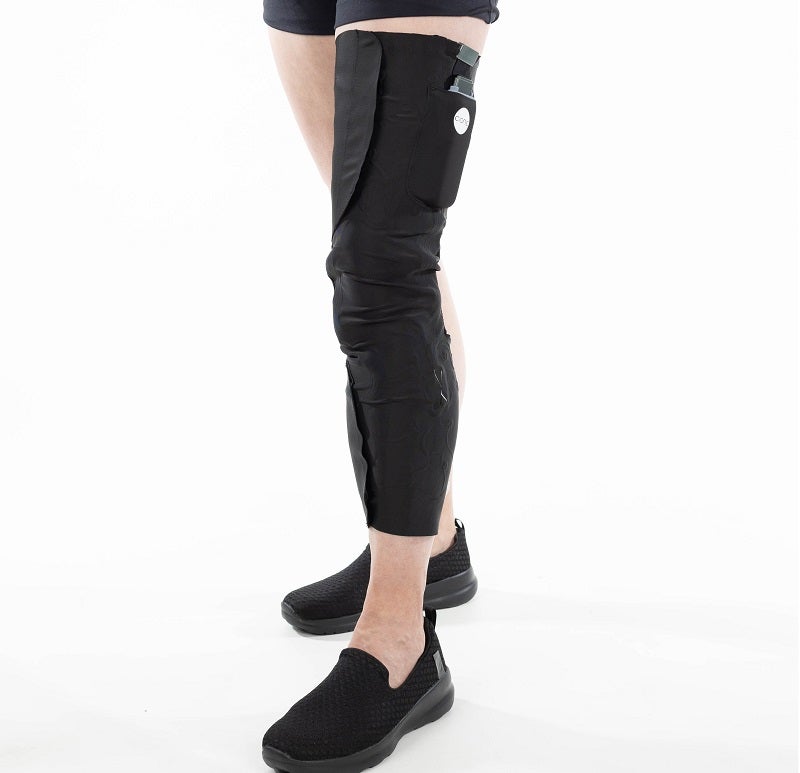
The US Food and Drug Administration (FDA) has granted approval for Cionic’s lightweight leg-worn Neural Sleeve.
The new wearable sleeve uses electrical stimulation to help improve the gait of people with mobility issues that are due to cerebral palsy (CP), multiple sclerosis (MS), stroke and other neurological conditions.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Cionic’s durable Neural Sleeve is designed for analysing, predicting and augmenting the movements of a person.
It uses advanced algorithms to read the signals sent to the muscles from the brain in real-time, predicting the user’s movement one tenth of a second before their foot lifts off the ground.
Using an array of sensors, the system measures body position and the way individual muscles fire during movement.
By measuring the electrical signal from the brain, the sensors can predict the person’s intended movement.
The device offers real-time augmentation and adjustment to a user’s movement.
It is made with soft, flexible fabric that is fitted to the leg of an individual and will also be available in various colours.
Cionic founder and CEO Jeremiah Robison said: “We are incredibly excited for the Cionic Neural Sleeve to be an FDA cleared Class II medical device and are ready to start impacting the lives of individuals with movement challenges in meaningful ways.
“Our mission is to become the new standard of care by addressing the underlying brain body connections, enabling confident movement and greater engagement in the community.”
After completing human factors and efficacy testing, the company submitted the device to obtain clearance from the FDA last November.
Furthermore, the company plans to expand into new areas, including conditions such as Parkinson’s, and other body parts.





