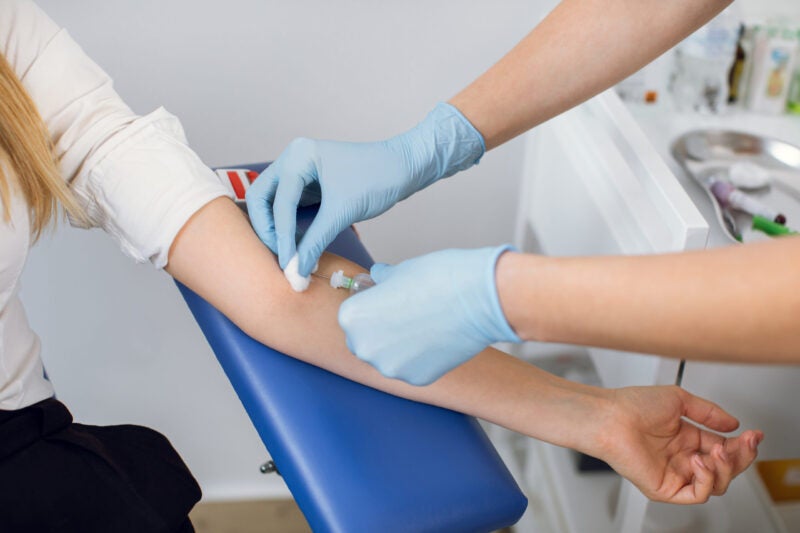
The US Food and Drug Administration (FDA) has granted 510(k) medical approval for SHOWA Group’s single-use nitrile gloves.
The company is the sole integrated manufacturer of nitrile, single-use personal protective equipment (PPE) gloves with full US onshore expertise.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
With the approval, SHOWA will now supply the market’s first biodegradable nitrile gloves approved for medical use.
Group purchasing organisations, hospitals and various healthcare buyers will be able to procure medical gloves that are certified to be safe for frontline workers.
The 510(k) clearance granted by the FDA extends across the SHOWA M7005PF single-use nitrile glove product line.
A Class I Medical device (glove), M7005PF is used in medical exams or any setting where the examiner and patient require protection from contamination.
It uses the Eco Best Technology (EBT) of the company and is completely biodegradable, breaking down by 82% in 386 days when analysed in a lab.
Further benefits and features of the product are its enhanced flexibility and dexterity, powder-free, exceptional tensile strength, no risk of natural rubber allergy, and resistance to leaks and tears.
In February, the company announced the expansion of its manufacturing site in Fayette, Alabama, US, which will boost its yearly domestic production capacity to 1.2 billion gloves by the end of the year.
The site expansion boosts the domestic supply of medical PPE in the US, lowering dependence on overseas PPE to guarantee that the country is better equipped for pandemics in the future.
SHOWA Group RAQA and technical manager Brian Moseley said: “SHOWA launched its biodegradable nitrile gloves a decade ago as an industry first, and since that time we’ve continued to invest in technology that reduces the environmental impact of PPE glove waste – a huge challenge with medical PPE during the pandemic.
“It can take 100 years or more for regular nitrile gloves to decompose in landfill conditions, but with SHOWA’s EBT, we’ve dramatically reduced decomposition time.”





