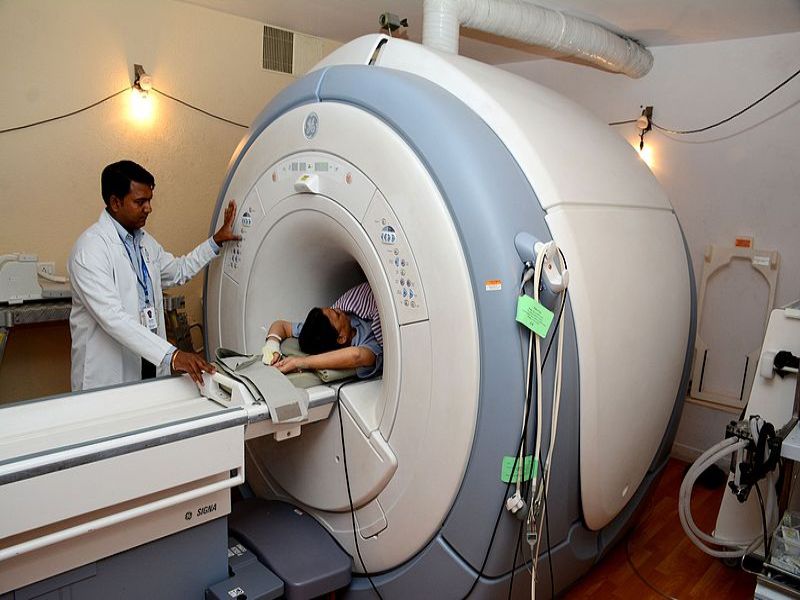
Axonics Modulation Technologies has received the US Food and Drug Administration (FDA) approval for its SmartMRI wireless patient Remote Control for the r-SNM System under a premarket approval (PMA) supplement.
The new remote control simplifies the full-body MRI process for patients. Prior to an MRI, an MRI technician can perform a simple check, using a patient’s remote control. This will spare patients the need to visit their implanting physician’s office or involving personnel from Axonics.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The company said that it will include the new patient remote control with SmartMRI technology in all new orders of the Axonics r-SNM System in the US, beginning this month.
Currently, the Axonics r-SNM System is approved for 1.5T full-body MRI scans in the US and both 1.5T and 3.0T full-body MRI scans in Canada and Europe.
The company submitted the submitted a PMA supplement to the FDA this year to obtain full-body MRI conditional labelling for 3.0T MR scanners in the US.
It expects the review to be completed towards the end of this year.
Axonics CEO Raymond Cohen said: “Delivering a superior experience to patients, physicians and their staff has been the key focus of our product development initiatives since Axonics’ founding. The introduction of this new patient remote control provides for significant convenience and reduces the burden on physician practices.
“This FDA approval follows the recent approval of both our next generation implantable neurostimulator and programmer and is part of a cadence of product enhancements that we are committed to pursuing.”
Last year, the company received the US FDA approval for r-SNM System to treat faecal incontinence. It also obtained the European CE-Mark approval for MRI conditional labelling of the r-SNM System.





