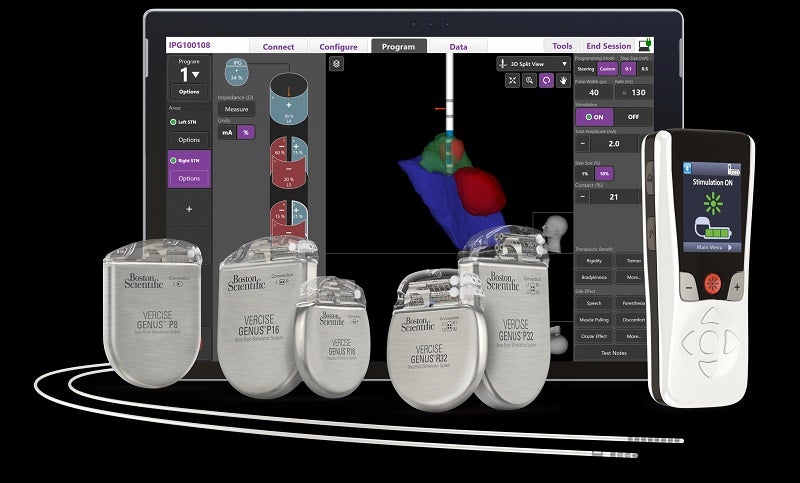
The US Food and Drug Administration (FDA) has granted approval to Boston Scientific’s Vercise Neural Navigator with STIMVIEW XT, the next-generation image-guided programming software for deep brain stimulation (DBS) therapy.
STIMVIEW XT has been developed in collaboration with software-driven medical technology company Brainlab.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
It allows clinicians to visualise real-time lead placement and stimulation modelling of brain anatomy for Parkinson’s disease or essential tremor patients.
An integrated visualisation software for DBS programming, STIMVIEW XT is used in combination with the Vercise Genus Deep Brain Stimulation portfolio.
It provides patient-specific 3D visualisation of brain anatomy for the personalisation of therapy.
The company stated that the Vercise Neural Navigator with STIMVIEW XT has been designed to minimise programming time, help localise lead placement and enable informed treatment.
Boston Scientific Neuromodulation senior vice-president Maulik Nanavaty said: “Every person’s experience living with Parkinson’s disease or essential tremor is unique, and their treatment should be as unique.
“Our technologies enable clinicians to precisely see, shape and steer DBS therapy to meet their patients’ individual needs.
“This latest advancement is a testament to how we’ll continue to deliver on meaningful innovations that support doctors.”
Using Boston Scientific visualisation software, clinicians in a recent study were able to adjust patient stimulation to an average of 20 minutes, reducing the programming time by 56%.
The company stated that the launch of the latest image-guided programming software in the US, which follows its launch in Europe, will provide broader access to clinicians treating Parkinson’s disease or essential tremor patients.
In January, Boston Scientific and GE Healthcare collaborated to provide an end-to-end interventional cardiac care portfolio in South-East Asia.





