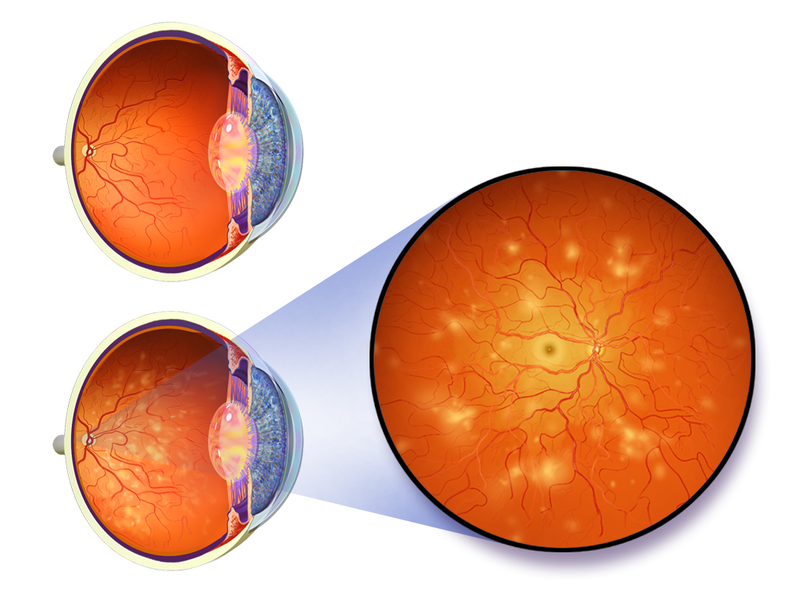
The US Food and Drug Administration (FDA) has approved the sale of an artificial intelligence (AI) based medical device for the detection of an eye disease associated with diabetes called diabetic retinopathy.
Reported to be the first device to provide screening decision without the need for a clinician, the IDx-DR software programme is designed to identify greater than a mild level of the condition in adults.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The software is installed on a cloud server and employs an AI algorithm to examine the uploaded eye images that are captured using the Topcon NW400 retinal camera.
If the images show more than mild diabetic retinopathy, the software refers to an eye care professional for further assessments or suggests a rescreen after 12 months in case of negative results for more than a mild version of the condition.
FDA Center for Devices and Radiological Health Ophthalmic, and Ear, Nose and Throat Devices division director Malvina Eydelman said: “Early detection of retinopathy is an important part of managing care for the millions of people with diabetes, yet many patients with diabetes are not adequately screened for diabetic retinopathy since about 50% of them do not see their eye doctor on a yearly basis.
“Today’s decision permits the marketing of a novel AI technology that can be used in a primary care doctor’s office.”
The FDA decision is based on the findings from a clinical study that involved retinal images from 900 diabetes patients at ten primary care sites.
Data demonstrated that IDx-DR accurately identified more than mild diabetic retinopathy in 87.4% of cases and correctly detected 89.5% of subjects without more than a mild level of the disorder.





