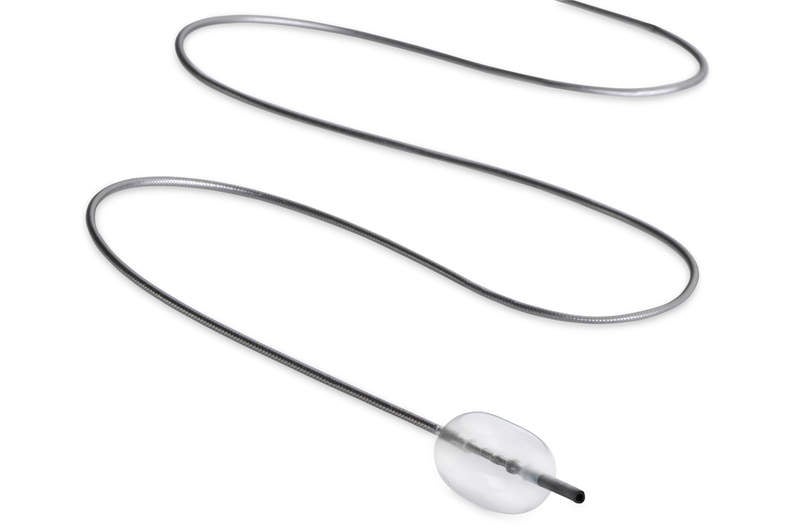
The US Food and Drug Administration (FDA) has granted 510(k) clearance to medical device company Embolx’s pressure-directed arterial embolisation system under the new Sniper Balloon Occlusion Microcatheters range.
The next-generation Sniper devices are intended for enhanced access and to facilitate easy navigation through small complex vascular structures.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Available in 110cm, 130cm and 150cm length variants, the microcatheter can be used to access femoral or radial arterial sites.
The company also added certain advancements to the balloon and made the tip design atraumatic in order to allow occlusion in large vessels and improve the device’s tracking ability inside the vessels.
Embolx president and CEO Michael Allen said: “Our next generation devices incorporate experience from more than 1,000 Sniper balloon occlusion microcatheters used to date.
“Now, interventional radiologists can do everything they would do with standard microcatheters, but with the added benefits of balloon occlusion. This is a big step forward and provides physicians with the most advanced transarterial delivery system for tumour and prostate treatment.”
The Sniper microcatheter is designed to control pressure in order to change blood flow‐dynamics and increase the delivery of therapeutic agents into target areas.
Currently, the device is used to treat cancerous tumours in various organs, including the liver, enlarged prostate (benign prostatic hyperplasia) and uterine fibroids.
The embolisation therapy is said to facilitate the delivery of drugs and embolic agents to the targeted treatment areas and at the same time protects surrounding healthy tissues.





