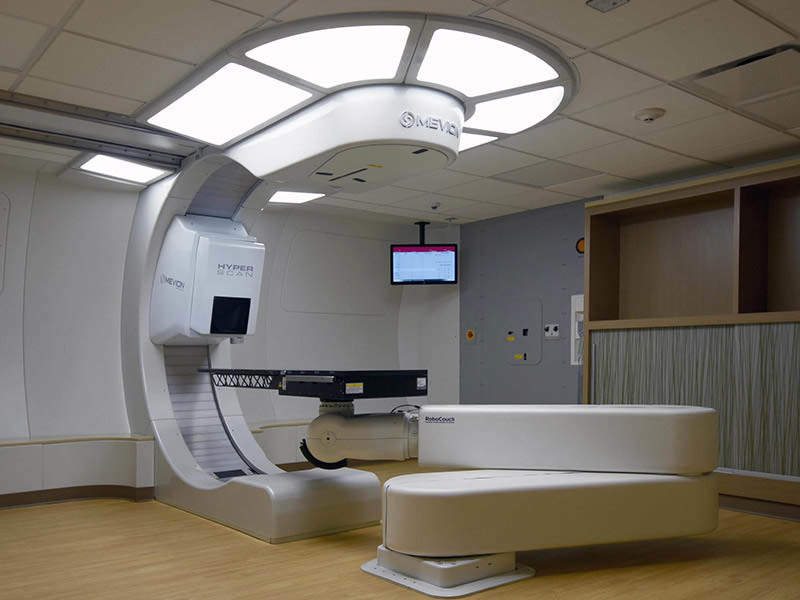
Mevion Medical Systems has obtained 510(k) clearance from the US Food and Drug Administration (FDA) for its MEVION S250i Proton Therapy System to deliver conformal radiation therapy treatment.
The compact system features Hyperscan pencil beam scanning (PBS) technology that provides energy layer switching and automated collimation systems for enabling rapid and sharp proton treatments.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Hyperscan PBS’ compact beam delivery path is said to minimise delivery times to lower than five seconds for certain fields, leading to decreased treatment errors, compared to other PBS technologies.
The PBS also uses the Adaptive Aperture proton multi-leaf collimator (pMLC), which is a robotically controlled collimation system with the ability to trim beam edges at every delivery layer.
This capability of the collimator is claimed to result in three times sharper radiation drop off at the delivery field edge, improving sparing of healthy tissue and limiting unnecessary radiation to sensitive locations.
Mevion Medical Systems CEO Joseph Jachinowski said: “The S250i system represents the next generation of intensity modulated proton therapy (IMPT) delivered in the most compact proton therapy platform.
“This is a very important milestone in our efforts to make precision proton therapy available to more patients in the fight against cancer.”
The firm has installed the first MEVION S250i Proton Therapy System at MedStar Georgetown University Hospital in Washington DC, US, to be offered as part of the hospital’s suite of cancer care offerings.





