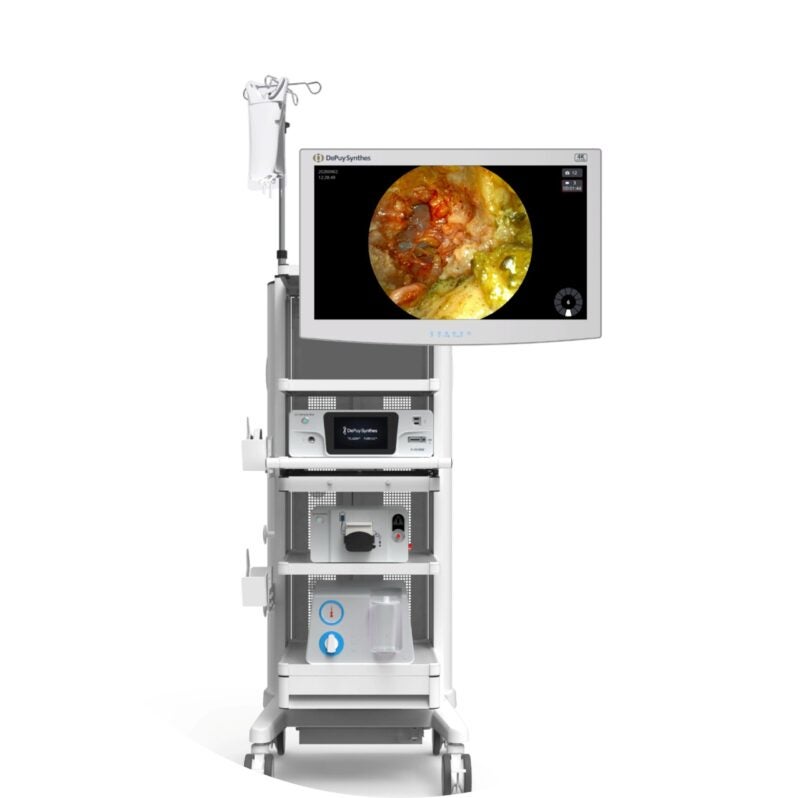
The US Food and Drug Administration (FDA) has granted 510(k) clearance for DePuy Synthes’ TELIGEN System, a combined technology platform.
Using digital tools for access and view, TELIGEN facilitates less invasive surgical transforaminal lumbar interbody fusion (MIS-TLIF) procedures.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The system includes a tower that offers various technologies, including a VueLIF-T Procedure Kit with a disposable HD camera, a camera control system, a TELIGEN Clear Discectomy Device and disposable ports.
The digitally equipped TELIGEN VUE Camera of the platform removes the requirement for a microscope and can offer an unhindered visualisation of the surgical site.
It enables multidirectional, expanded and hands-free view during the procedure.
Surgeons could adjust the image clarity as per their preference using the self-cleaning camera, which also has LED lighting.
Additionally, the system merges with the UNLEASH bundle of implant solutions, which is created to simplify the key MIS-TLIF stages.
As against current MIS technologies, the TELIGEN System could be efficient and cost-effective for hospitals.
The system was demonstrated to lower fluoroscopy time by 47% versus MIS-TLIF procedures carried out using a surgical microscope in a cadaveric study.
The company anticipates making the system available in the country later this year.
DePuy Synthes Spine worldwide president Russell Powers said: “Improving the MIS spinal surgery experience for both patients and surgeons is a critical step to addressing unmet needs in the industry.
“With our groundbreaking TELIGEN Technology Platform, we’re providing a better field of view to help improve patient care and increase efficiencies.”
In November last year, the company introduced the UNIUM System for small bone, sports medicine, spine and thorax procedures.



