The US Food and Drug Administration (FDA) has granted 510(k) clearance to a new swallowable balloon device called EsoCheck for the detection of pre-cancerous Barrett’s oesophagus.
EsoCheck is part of a test developed by researchers at the Case Western Reserve University School of Medicine and University Hospitals Cleveland Medical Center to detect the disease prematurely.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
In addition to the pill-sized, swallowable device designed to obtain oesophagus samples, the test comprises a DNA assay to identify Barrett’s oesophagus and oesophagal cancers.
The device has secured FDA clearance and is being manufactured by Lucid Diagnostics.
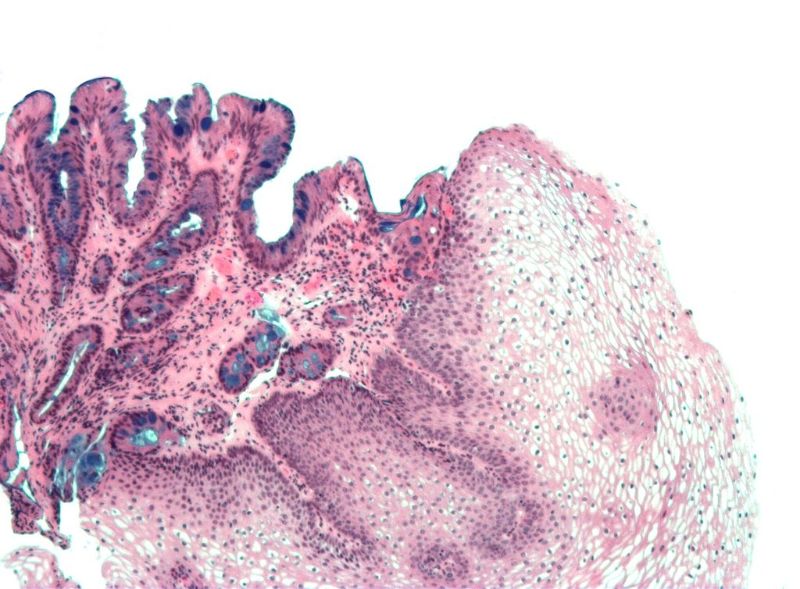
Barrett’s oesophagus is the precursor lesion of oesophagal adenocarcinoma, which is associated with high mortality but can be prevented with diagnosis and intervention at the precursor stage.
Endoscopy is commonly used to detect Barrett’s oesophagus. However, it is considered costly and invasive.
Researchers developed an outpatient test that requires five minutes to detect the condition and is said to be more than 90% sensitive.
During the test, patients have to swallow a capsule attached to a silicone catheter. After reaching the stomach, the small balloon inside the capsule inflates by injection of air via the catheter.
The balloon is then used to swab the lower oesophagus, where Barrett’s oesophagus originates. Subsequently, the balloon is deflated and sent back into the capsule to protect the sample.
Following the retrieval of the capsule, DNA is obtained from the balloon surface and analysed for aberrant DNA methylation related to the condition.
Case Western Reserve University School of Medicine faculty Amitabh Chak said: “FDA clearance of the EsoCheck device means the device is now available to patients and physicians as a non-endoscopic method for sampling the oesophagus. This will make it much easier to detect Barrett’s oesophagus in the outpatient setting.”
In an 86-patient clinical trial, the balloon device was found to be well-tolerated.
Meanwhile, separate research at the School of Medicine used genome-wide sequencing and detected that the VIM and CCNA1 genes get chemically modified by DNA methylation in Barrett’s oesophagus.
A study of more than 300 oesophagus endoscopy samples showed the DNA test to be 90% accurate in detecting the condition or identifying a normal oesophagus.
Rresearchers intend to partner with Lucid Diagnostics to offer the VIM and CCNA1 DNA assays to test the DNA extracted using the EsoCheck device.





