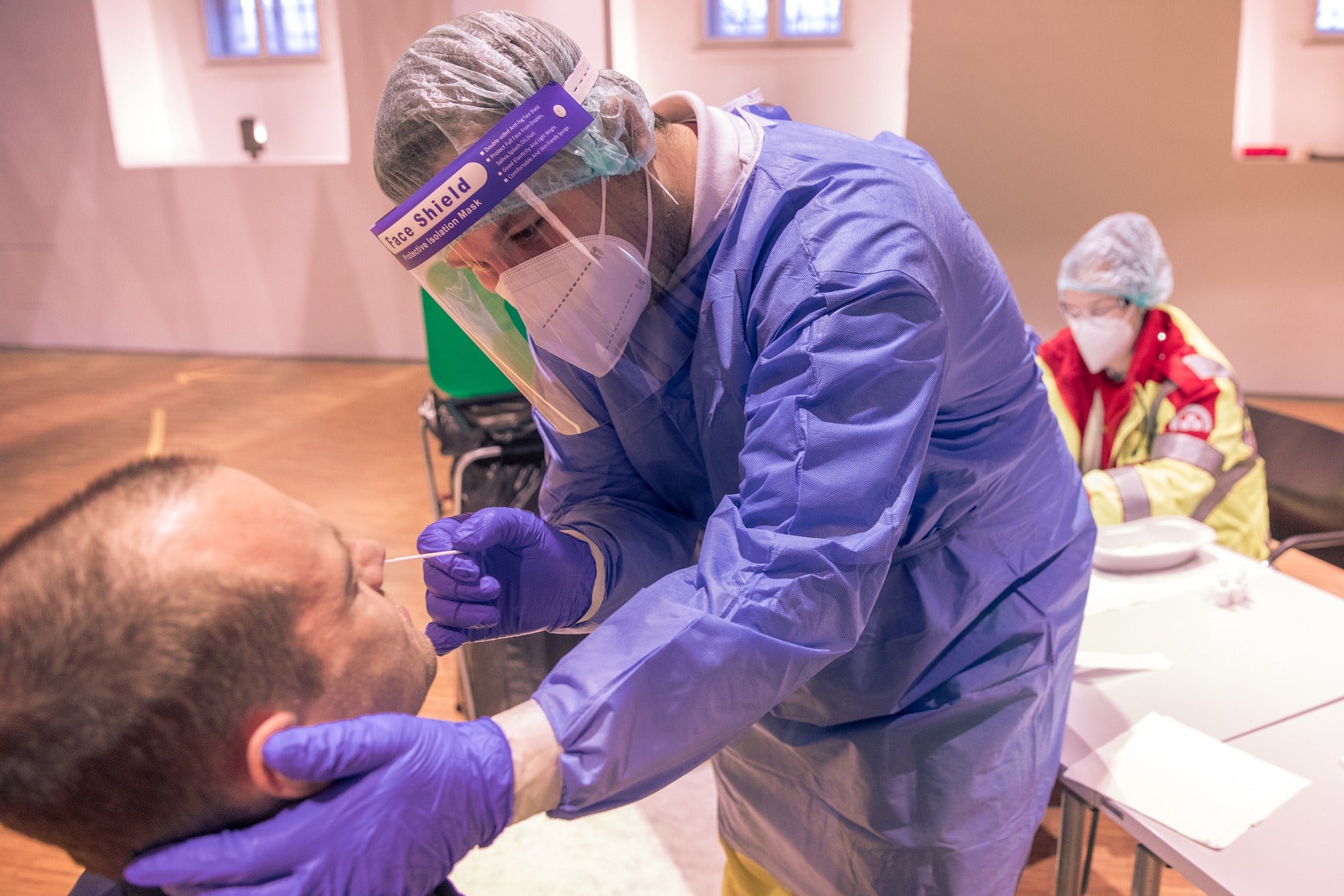
The US Food and Drug Administration (FDA) has granted emergency use authorization (EUA) for Applied BioCode’s CoV-2 Flu Plus Assay.
The new multiplex molecular diagnostic BioCode CoV-2 Flu Plus Assay is designed to detect and differentiate between SARS-CoV-2, respiratory syncytial virus (RSV), as well as influenza A subtypes such as seasonal H1, 2009 H1N1, H3, Influenza B simultaneously.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
It is intended to aid physicians to evaluate the clinical signs and symptoms of patients with a respiratory infection.
The PCR-based test uses nasopharyngeal swab specimens to detect SARS-CoV-2, influenza A, influenza B and RSV.
The assay runs on the company’s BioCode MDx-3000 Molecular System, which allows clinical laboratories to provide up to 564 results a day.
It represents Applied BioCode’s third EUA authorisation for Covid-19 testing from the FDA.
Previously, the company’s BioCode SARS CoV-2 Assay received EUA and for pooling up to five samples for the same test.
Applied BioCode president Winston Ho said: “We have taken an important next step in strengthening our infectious disease menu by upgrading the BioCode SARS-CoV-2 test to a panel that now also includes influenza A, influenza B and RSV.
“Our BioCode CoV-2 Flu Plus Assay will not only increase laboratory and hospital efficiency but will also help physicians to make a more informed decision.”
The new diagnostic assay builds on the company’s existing portfolio of syndromic assays that include its stand-alone EUA authorized BioCode SARS-CoV-2 Assay, FDA 510(k) cleared BioCode Gastrointestinal Pathogen Panel, and FDA 510(k) cleared BioCode Respiratory Pathogen Panel.
In November, Meridian Bioscience secured EUA from the US FDA for its Revogene SARS-CoV-2 assay.





