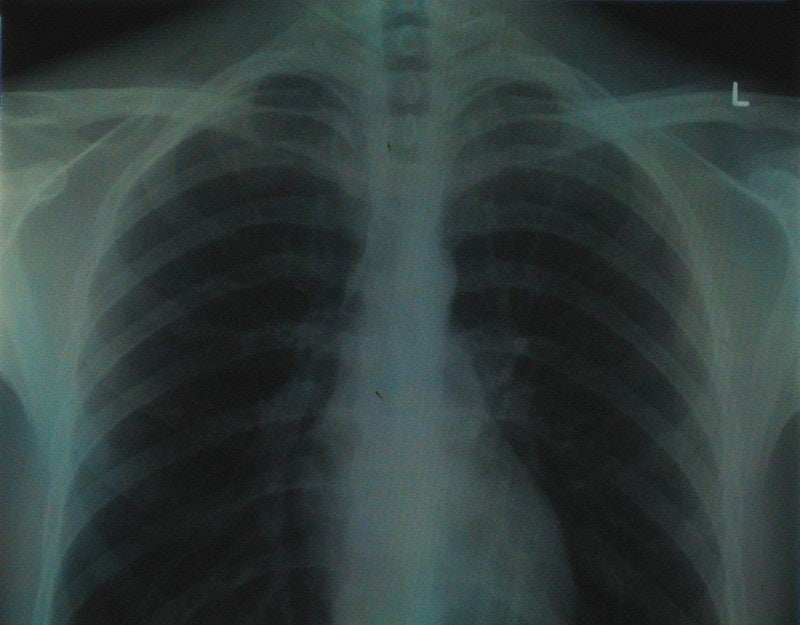
The Food and Drug Administration (FDA) has granted 510(k) clearance to health tech firm Qure.ai’s artificial intelligence (AI) algorithm, known as qXR-BT.
The new algorithm is capable of automating the manual measurement process for endotracheal as well as tracheostomy tubes and helps clinicians assess Breathing Tube (BT) placement.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
It uses chest X-rays of intubated patients to assist clinicians in identifying BT placement and automating measurements.
Qure.ai CEO and co-founder Prashant Warier said: “We are pleased to have received FDA clearance for qXR-BT. In the last two years, we have seen the need to decrease processing times and solve workflow delays.
“Especially in the wake of the Covid-19 pandemic and the need for mechanical ventilation in affected patients, the need for prompt assistance to an overburdened healthcare workforce is paramount.”
BT position is analysed by the qXT-BT algorithm. A report with automated measurements and the positional accuracy of the tube is provided for the physician in less than one minute.
With this, clinicians can quickly identify tube positions and decide if additional attention is needed.
The vendor-agnostic algorithm has been designed to work on both stationary X-ray and portable machines.
Harvard Medical School Radiology professor Dr Mannudeep Kalra was involved in a research collaboration that assessed the technology.
Kalra said: “Daily monitoring of tubes is critical for all intubated inpatients, and sometimes an arduous task on the portable exam with either the carina obscured or the tip not visible.
“An accurate AI solution could be a valuable aid for reporting on these chest X-rays – especially with the measurement.”





