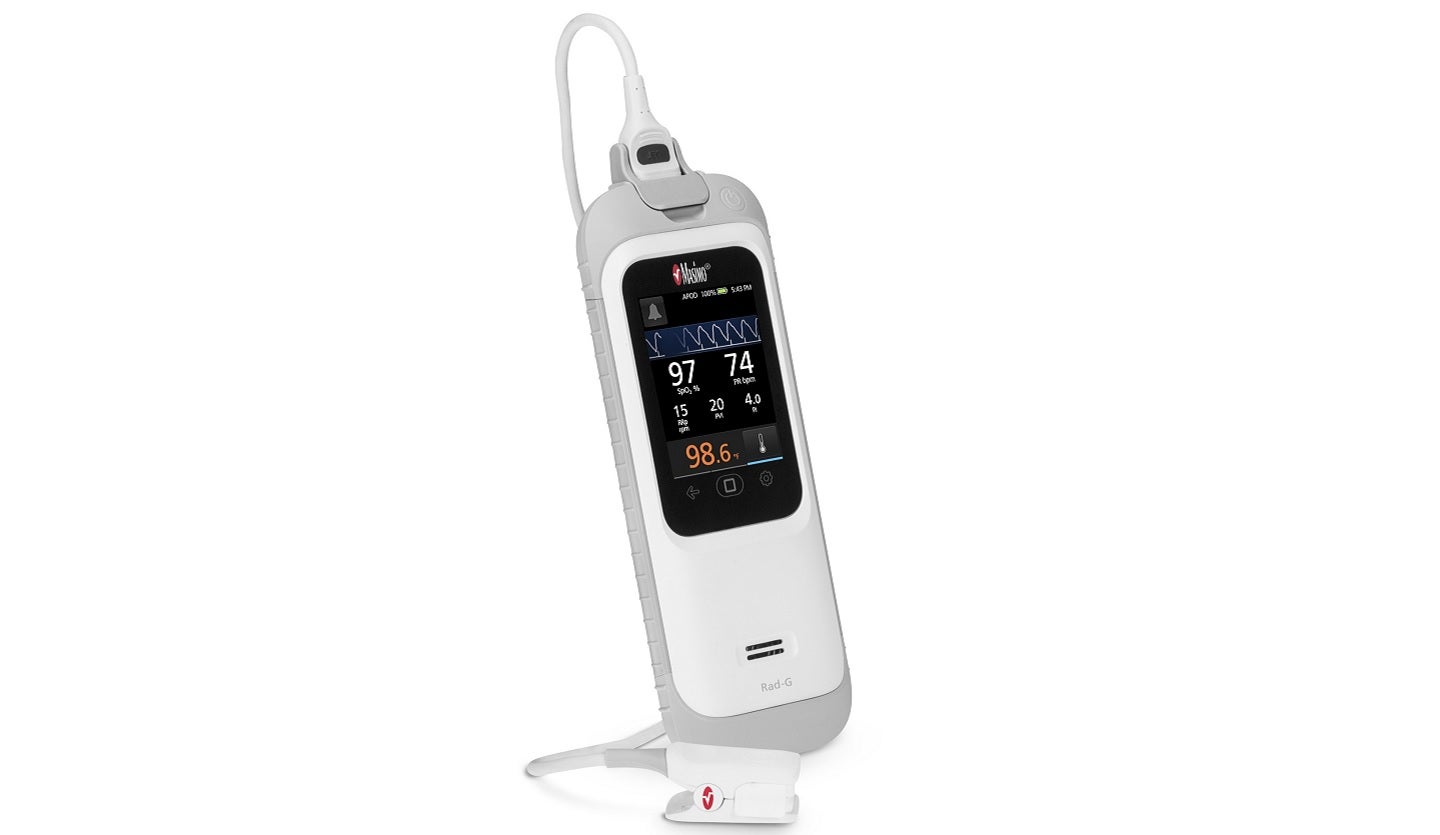
The US Food and Drug Administration (FDA) has granted 510(k) clearance to Masimo’s Rad-G with Temperature device for pulse oximetry and temperature measurement.
The Rad-G with Temperature is a rugged, two-in-one handheld monitor which provides SET pulse oximetry, respiration rate from the pleth (RRp) and other key parameters.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The company initially developed the device as a spot-check device in collaboration with The Bill and Melinda Gates Foundation for use in pneumonia screening. The device was first introduced outside the US.
The lightweight device comes with an integrated non-invasive forehead thermometry, a sturdy rubber casing and a long-lasting rechargeable battery.
It is designed to measure several physiological parameters, including pleth variability index, pulse rate, oxygen saturation (SpO2), RRp, temperature and perfusion index to support clinical management.
The device can be used in various settings including wellness clinics, first-responder scenarios, outpatient services, physicians’ offices and urgent care facilities.
Masimo founder and CEO Joe Kiani said: “With Rad-G, we set out to create an accessible, high-quality care solution that could be used in a multitude of care settings to serve the five billion people on our planet that to date have not had access to pulse oximetry, let alone SET pulse oximetry.
“With the addition of temperature measurements, Rad-G is more versatile than ever, streamlining the assessment of multiple key vital signs.
“With this FDA clearance, Rad-G with Temperature can now be deployed across the US in addition to many other parts of the world, helping support clinicians in almost any care scenario.”
Rad-G with Temperature comprises a non-contact thermometer which does not require probe covers or other disposable accessories.
With one-touch operation, the thermometer facilitates easy and effective temperature screening along with oxygen saturation and respiration rate.
The device uses Masimo SET Measure-through Motion and Low Perfusion pulse oximetry to offer SpO2 and PR monitoring.
It is also compatible with the broad portfolio of the company’s single-patient-use adhesive sensors, including Masimo RD SET sensors.





