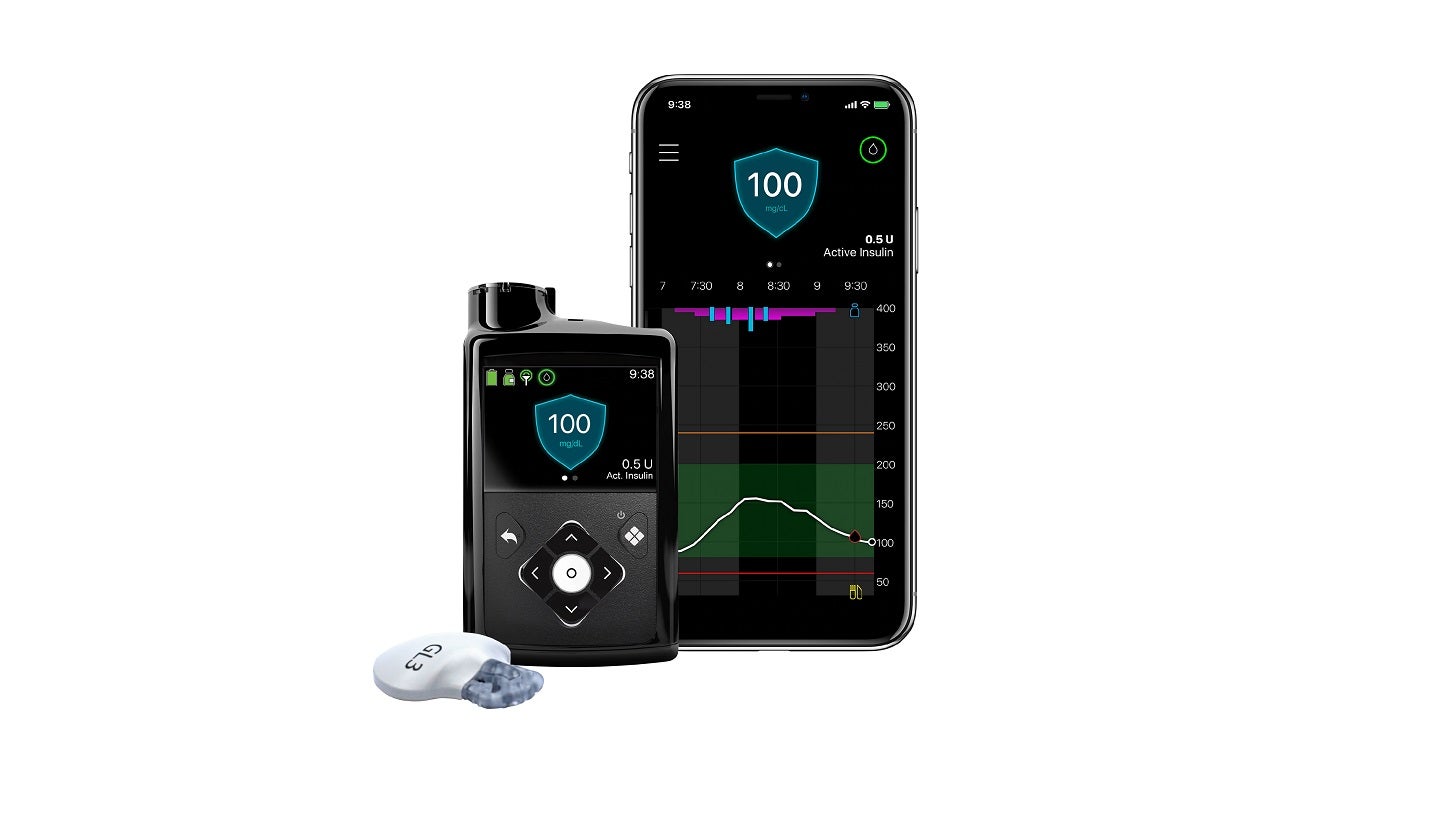
The US Food and Drug Administration (FDA) has granted approval for Medtronic’s insulin pump, the MiniMed 780G system.
The new insulin pump system comes with the Guardian 4 sensor which eliminates the need for finger pricks while in SmartGuard mode.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
It is claimed to be the only FDA-approved insulin pump system with meal-detection technology that automatically adjusts and corrects glucose levels every five minutes for basal (background) and bolus (mealtime) insulin needs.
The MiniMed 780G system facilitates the lowest glucose target setting where users can set as low as a 100mg/dL dose in any automated insulin pump currently available in the market.
This setting closely mimics the average glucose delivery of people without diabetes and will automatically adjust and correct basal insulin based on the set target.
Medtronic Diabetes president and executive vice-president Que Dallara said: “Mealtimes prove to be one of the biggest challenges for people living with type 1 diabetes and now for the first time, the MiniMed 780G system addresses this unmet need with automatic, real-time insulin corrections.
“A lot can happen to blood sugars in the span of an hour or even just a few minutes, so we’ve designed our system for real life – the algorithm adapts to the user and helps compensate for everyday challenges that are quite common around mealtimes… we’re excited to deliver a system with ease of use at the forefront.”





