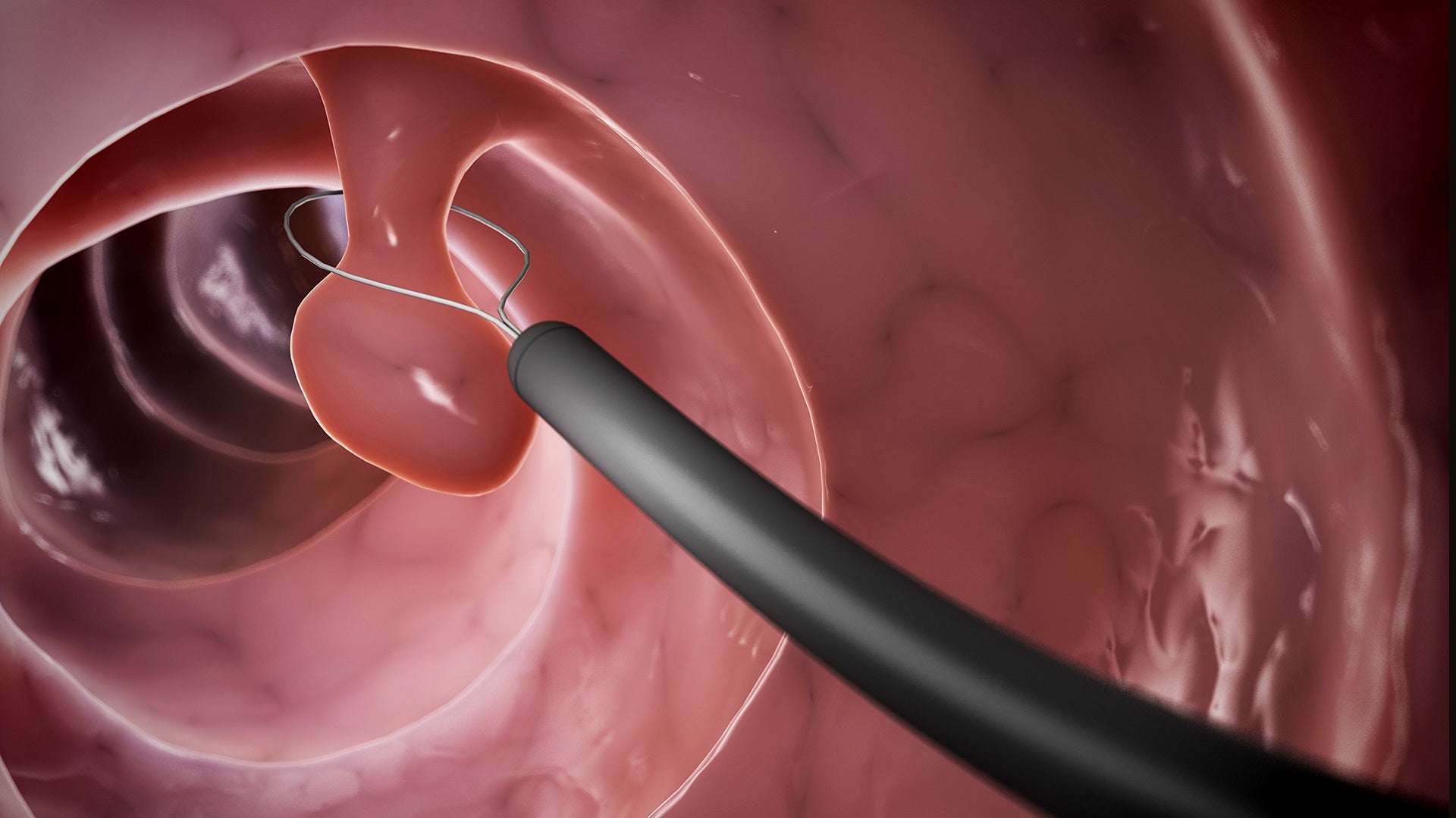
The US Food and Drug Administration (FDA) has granted 510(k) clearance to Olympus’ Narrow Band Imaging (NBI) to assess the neoplastic potential of colorectal polyps.
NBI is an optical imaging technology that improves the visibility of vessels and surface patterns on the mucosa.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Physicians can make high-confidence predictions of histology for polyps 5mm or smaller, called diminutive polyps, by applying the NBI International Colorectal Endoscopic (NICE) classification during a screening colonoscopy.
Olympus America Endoscopy vice-president Kevin Mancini said: “We are excited about these newly cleared indications for using NBI to support colorectal cancer screening.
“NBI is an important tool, available as standard on all Olympus colonoscopes, which can be used by physicians to assist in decision making with the goal of improving patient care.”
In support of the clearance, the company submitted data that included a meta-analysis of prospective real-time clinical studies of NBI use at the time of colonoscopy.
The data showed endoscopists using the NICE classification demonstrated 93% sensitivity, 85% specificity and more than 90% negative predictive value in predicting adenomatous histology of diminutive polyps during colonoscopy with high confidence.
Additionally, they have achieved more than 90% agreement with pathological analysis in assigning post-polypectomy patient surveillance intervals after colonoscopy.
In a randomised clinical trial, endoscopists using near focus mode colonoscopes were more likely to make high confidence predictions of diminutive polyp histology than those using the standard focus colonoscopies.
Olympus noted that at the time of colonoscopy, polyps are usually detected and removed and sent out for pathological diagnosis.
NBI, which can filter out all but blue and green lights absorbed by haemoglobin, also has clinical applications in urology, pulmonology and rhinolaryngology.



